Antiepileptic Stiripentol May Influence Bones
Abstract
:1. Introduction
2. Results
2.1. Body Weight
2.2. Serum Parameters
2.3. Bone Macrometric Parameters
2.4. Bone Mineral Density
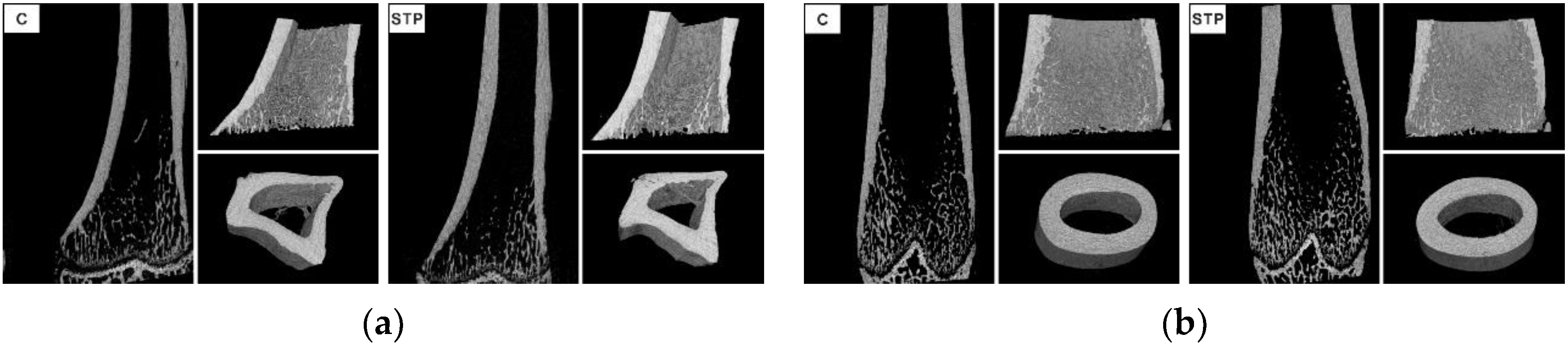
2.5. Micro X-ray Computed Tomography
2.6. Three-Point Bending Test
3. Discussion
4. Materials and Methods
4.1. Ethical Statements and Animals
4.2. Design of the Study
4.3. Serum Parameters
4.4. Macrometric Measurements
4.5. Densitometry
4.6. Micro X-ray Computed Tomography
4.7. Three-Point Bending Test
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Fan, H.-C.; Lee, H.-S.; Chang, K.-P.; Lee, Y.-Y.; Lai, H.-C.; Hung, P.-L.; Lee, H.-F.; Chi, C.-S. The Impact of Anti-Epileptic Drugs on Growth and Bone Metabolism. Int. J. Mol. Sci. 2016, 17, 1242. [Google Scholar] [CrossRef] [Green Version]
- Löscher, W.; Potschka, H.; Sisodiya, S.M.; Vezzani, A. Drug Resistance in Epilepsy: Clinical Impact, Potential Mechanisms, and New Innovative Treatment Options. Pharmacol. Rev. 2020, 72, 606–638. [Google Scholar] [CrossRef] [PubMed]
- Aneja, S.; Sharma, S. Newer anti-epileptic drugs. Indian Pediatr. 2013, 50, 1033–1040. [Google Scholar] [CrossRef] [PubMed]
- Miziak, B.; Chrościńska-Krawczyk, M.; Czuczwar, S.J. An update on the problem of osteoporosis in people with epilepsy taking antiepileptic drugs. Expert Opin. Drug Saf. 2019, 18, 679–689. [Google Scholar] [CrossRef]
- Valsamis, H.A.; Arora, S.K.; Labban, B.; McFarlane, S.I. Antiepileptic drugs and bone metabolism. Nutr. Metab. 2006, 3, 36. [Google Scholar] [CrossRef] [Green Version]
- Shen, C.; Chen, F.; Zhang, Y.; Guo, Y.; Ding, M. Association between use of antiepileptic drugs and fracture risk: A systematic review and meta-analysis. Bone 2014, 64, 246–253. [Google Scholar] [CrossRef]
- Fekete, S.; Simko, J.; Gradosova, I.; Malakova, J.; Zivna, H.; Palicka, V.; Zivny, P. The effect of levetiracetam on rat bone mass, structure and metabolism. Epilepsy Res. 2013, 107, 56–60. [Google Scholar] [CrossRef]
- Aksoy, D.; Güveli, B.T.; Ak, P.D.; Sarı, H.; Ataklı, D.; Arpacı, B. Effects of Oxcarbazepine and Levetiracetam on Calcium, Ionized Calcium, and 25-OH Vitamin-D3 Levels in Patients with Epilepsy. Clin. Psychopharmacol. Neurosci. 2016, 14, 74–78. [Google Scholar] [CrossRef] [Green Version]
- Chiron, C. Current therapeutic procedures in Dravet syndrome. Dev. Med. Child Neurol. 2011, 53, 16–18. [Google Scholar] [CrossRef] [PubMed]
- Nickels, K.C.; Wirrell, E.C. Stiripentol in the Management of Epilepsy. CNS Drugs 2017, 31, 405–416. [Google Scholar] [CrossRef] [PubMed]
- Łuszczki, J.J. Third-generation antiepileptic drugs: Mechanisms of action, pharmacokinetics and interactions. Pharmacol. Rep. 2009, 61, 197–216. [Google Scholar] [CrossRef]
- Verleye, M.; Buttigieg, D.; Steinschneider, R. Neuroprotective activity of stiripentol with a possible involvement of voltage-dependent calcium and sodium channels. J. Neurosci. Res. 2016, 94, 179–189. [Google Scholar] [CrossRef]
- Mattson, R.H.; Gidal, B.E. Fractures, epilepsy, and antiepileptic drugs. Epilepsy Behav. 2004, 5, 36–40. [Google Scholar] [CrossRef] [PubMed]
- Patsalos, P.N. Drug Interactions with the Newer Antiepileptic Drugs (AEDs)—Part 1: Pharmacokinetic and Pharmacodynamic Interactions between AEDs. Clin. Pharmacokinet. 2013, 52, 927–966. [Google Scholar] [CrossRef]
- Wheless, J.W.; Fulton, S.P.; Mudigoudar, B.D. Dravet Syndrome: A Review of Current Management. Pediatr. Neurol. 2020, 107, 28–40. [Google Scholar] [CrossRef] [PubMed]
- Strzelczyk, A.; Schubert-Bast, S. Therapeutic advances in Dravet syndrome: A targeted literature review. Expert Rev. Neurother. 2020, 20, 1065–1079. [Google Scholar] [CrossRef]
- Selvarajah, A.; Zulfiqar-Ali, Q.; Marques, P.; Rong, M.; Andrade, D.M. A systematic review of adults with Dravet syndrome. Seizure 2021, 87, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Verbeek, N.E.; Van Kempen, M.; Gunning, W.B.; Renier, W.O.; Westland, B.; Lindhout, D.; Brilstra, E.H. Adults with a history of possible Dravet syndrome: An illustration of the importance of analysis of the SCN1A gene. Epilepsia 2011, 52, e23–e25. [Google Scholar] [CrossRef]
- Samaniego, E.; Sheth, R.D. Bone Consequences of Epilepsy and Antiepileptic Medications. Semin. Pediatr. Neurol. 2007, 14, 196–200. [Google Scholar] [CrossRef]
- Eschbach, K.; Scarbro, S.; Juarez-Colunga, E.; Allen, V.; Hsu, S.; Knupp, K. Growth and endocrine function in children with Dravet syndrome. Seizure 2017, 52, 117–122. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huttunen, M.M.; Tillman, I.; Viljakainen, H.T.; Tuukkanen, J.; Peng, Z.; Pekkinen, M.; Lamberg-Allardt, C.J. High Dietary Phosphate Intake Reduces Bone Strength in the Growing Rat Skeleton. J. Bone Miner. Res. 2007, 22, 83–92. [Google Scholar] [CrossRef]
- Hernández-Becerra, E.; Gutiérrez-Cortez, E.; Del Real, A.; Rojas-Molina, A.; Rodríguez-García, M.; Rubio, E.; Quintero-García, M.; Rojas-Molina, I. Bone Mineral Density, Mechanical, Microstructural Properties and Mineral Content of the Femur in Growing Rats Fed with Cactus Opuntia ficus indica (L.) Mill. (Cactaceae) Cladodes as Calcium Source in Diet. Nutrients 2017, 9, 108. [Google Scholar] [CrossRef] [Green Version]
- Jee, W.S.; Yao, W. Overview: Animal models of osteopenia and osteoporosis. J. Musculoskelet. Neuronal Interact. 2001, 1, 193–207. [Google Scholar] [PubMed]
- El-Haggar, S.M.; Mostafa, T.M.; Allah, H.M.S.; Akef, G.H. Levetiracetam and lamotrigine effects as mono- and polytherapy on bone mineral density in epileptic patients. Arq. Neuro Psiquiatr. 2018, 76, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Beniczky, S.; Viken, J.; Jensen, L.T.; Andersen, N.B. Bone mineral density in adult patients treated with various antiepileptic drugs. Seizure 2012, 21, 471–472. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanda, J.; Izumo, N.; Kobayashi, Y.; Onodera, K.; Shimakura, T.; Yamamoto, N.; Takahashi, H.E.; Wakabayashi, H. Effects of the antiepileptic drugs topiramate and lamotrigine on bone metabolism in rats. Biomed. Res. 2017, 38, 297–305. [Google Scholar] [CrossRef] [Green Version]
- Kanda, J.; Izumo, N.; Kobayashi, Y.; Onodera, K.; Shimakura, T.; Yamamoto, N.; Takahashi, H.E.; Wakabayashi, H. Effects of the Antiepileptic Drugs Phenytoin, Gabapentin, and Levetiracetam on Bone Strength, Bone Mass, and Bone Turnover in Rats. Biol. Pharm. Bull. 2017, 40, 1934–1940. [Google Scholar] [CrossRef] [Green Version]
- Takahashi, A.; Onodera, K.; Kamei, J.; Sakurada, S.; Shinoda, H.; Miyazaki, S.; Saito, T.; Mayanagi, H. Effects of Chronic Administration of Zonisamide, an Antiepileptic Drug, on Bone Mineral Density and Their Prevention with Alfacalcidol in Growing Rats. J. Pharmacol. Sci. 2003, 91, 313–318. [Google Scholar] [CrossRef] [Green Version]
- Nowińska, B.; Folwarczna, J.; Dusiło, A.; Pytlik, M.; Sliwiński, L.; Cegieła, U.; Kaczmarczyk-Sedlak, I.; Pietryka, W.; Hanke, T.; Trzeciak, H.I. Effects of vigabatrin on the skeletal system of young rats. Acta Pol. Pharm. 2012, 69, 327–334. [Google Scholar]
- Peacock, M. Phosphate Metabolism in Health and Disease. Calcif. Tissue Int. 2021, 108, 3–15. [Google Scholar] [CrossRef]
- Bouillon, R.; Marcocci, C.; Carmeliet, G.; Bikle, D.; White, J.H.; Dawson-Hughes, B.; Lips, P.; Munns, C.F.; Lazaretti-Castro, M.; Giustina, A.; et al. Skeletal and Extraskeletal Actions of Vitamin D: Current Evidence and Outstanding Questions. Endocr. Rev. 2019, 40, 1109–1151. [Google Scholar] [CrossRef] [Green Version]
- Vervloet, M.G.; van Ballegooijen, A.J. Prevention and treatment of hyperphosphatemia in chronic kidney disease. Kidney Int. 2018, 93, 1060–1072. [Google Scholar] [CrossRef]
- Liu, Y. Cellular and molecular mechanisms of renal fibrosis. Nat. Rev. Nephrol. 2011, 7, 684–696. [Google Scholar] [CrossRef] [PubMed]
- Marcinowska-Suchowierska, E.; Kupisz-Urbanska, M.; Lukaszkiewicz, J.; Pludowski, P.; Jones, G. Vitamin D Toxicity—A clinical perspective. Front. Endocrinol. 2018, 9, 550. [Google Scholar] [CrossRef] [Green Version]
- Hamed, S. The effect of antiepileptic drugs on the kidney function and structure. Expert Rev. Clin. Pharmacol. 2017, 10, 993–1006. [Google Scholar] [CrossRef]
- Heidari, R.; Jafari, F.; Khodaei, F.; Shiraziyeganeh, B.; Niknahad, H. Mechanism of valproic acid-induced Fanconi syndrome involves mitochondrial dysfunction and oxidative stress in rat kidney. Nephrology 2018, 23, 351–361. [Google Scholar] [CrossRef]
- Hamed, S.A.; Rageh, T.A.; Mohamad, A.O.; Elnour, S.M.A.; Nour, S.E. Renal dysfunctions/injury in adult epilepsy patients treated with carbamazepine or valproate. Expert Rev. Clin. Pharmacol. 2018, 11, 819–824. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Huang, J. Drug-Induced Nephrotoxicity: Pathogenic Mechanisms, Biomarkers and Prevention Strategies. Curr. Drug Metab. 2018, 19, 559–567. [Google Scholar] [CrossRef]
- Joyce, E.L.; Kane-Gill, S.L.; Fuhrman, D.Y.; Kellum, J.A. Drug-associated acute kidney injury: Who’s at risk? Pediatr. Nephrol. 2016, 32, 59–69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Griffin, B.R.; Faubel, S.; Edelstein, C.L. Biomarkers of drug-induced kidney toxicity. Ther. Drug Monit. 2019, 41, 213–226. [Google Scholar] [CrossRef]
- Beker, B.M.; Corleto, M.G.; Fieiras, C.; Musso, C.G. Novel acute kidney injury biomarkers: Their characteristics, utility and concerns. Int. Urol. Nephrol. 2018, 50, 705–713. [Google Scholar] [CrossRef] [PubMed]
- Waring, W.S.; Moonie, A. Earlier recognition of nephrotoxicity using novel biomarkers of acute kidney injury. Clin. Toxicol. 2011, 49, 720–728. [Google Scholar] [CrossRef] [PubMed]
- Desanti De Oliveira, B.; Xu, K.; Shen, T.H.; Callahan, M.; Kiryluk, K.; D’Agati, V.D.; Tatonetti, N.P.; Barasch, J.; Devarajan, P. Molecular nephrology: Types of acute tubular injury. Nat. Rev. Nephrol. 2019, 15, 599–612. [Google Scholar] [CrossRef] [PubMed]
- Haase, M.; Bellomo, R.; Devarajan, P.; Schlattmann, P.; Haase-Fielitz, A. Accuracy of Neutrophil Gelatinase-Associated Lipocalin (NGAL) in Diagnosis and Prognosis in Acute Kidney Injury: A Systematic Review and Meta-Analysis. Am. J. Kidney Dis. 2009, 54, 1012–1024. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, W.; Li, Z.; Chen, Y.; Wu, H.; Zhang, S.; Chen, X. Prediction Value of Serum NGAL in the Diagnosis and Prognosis of Experimental Acute and Chronic Kidney Injuries. Biomolecules 2020, 10, 981. [Google Scholar] [CrossRef] [PubMed]
- Rysz, J.; Gluba-Brzózka, A.; Franczyk, B.; Jablonowski, Z.; Cialkowska-Rysz, A. Novel Biomarkers in the Diagnosis of Chronic Kidney Disease and the Prediction of Its Outcome. Int. J. Mol. Sci. 2017, 18, 1702. [Google Scholar] [CrossRef]
- Mitsnefes, M.M.; Kathman, T.S.; Mishra, J.; Kartal, J.; Khoury, P.R.; Nickolas, T.L.; Barasch, J.; Devarajan, P. Serum neutrophil gelatinase-associated lipocalin as a marker of renal function in children with chronic kidney disease. Pediatr. Nephrol. 2007, 22, 101–108. [Google Scholar] [CrossRef]
- Zhang, W.R.; Parikh, C.R. Biomarkers of Acute and Chronic Kidney Disease. Ann. Rev. Physiol. 2019, 81, 309–333. [Google Scholar] [CrossRef]
- Eller-Vainicher, C.; Falchetti, A.; Gennari, L.; Cairoli, E.; Bertoldo, F.; Vescini, F.; Scillitani, A.; Chiodini, L. Diagnosis of Endocrine Disease: Evaluation of bone fragility in endocrine disorders. Eur. J. Endocrinol. 2019, 180, R213–R232. [Google Scholar] [CrossRef] [Green Version]
- Gois, P.H.F.; Wolley, M.; Ranganathan, D.; Seguro, A.C. Vitamin D Deficiency in Chronic Kidney Disease: Recent Evidence and Controversies. Int. J. Environ. Res. Public Health 2018, 15, 1773. [Google Scholar] [CrossRef] [Green Version]
- Mohamad, N.V.; Soelaiman, I.N.; Chin, K.Y. A concise review of testosterone and bone health. Clin. Interv. Aging 2016, 11, 1317–1324. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.F.; Lin, P.W.; Tsai, Y.R.; Yang, Y.C.; Kang, H.Y. Androgens and Androgen Receptor Actions on Bone Health and Disease: From Androgen Deficiency to Androgen Therapy. Cells 2019, 8, 1318. [Google Scholar] [CrossRef] [Green Version]
- Svalheim, S.; Sveberg, L.; Mochol, M.; Taubøll, E. Interactions between antiepileptic drugs and hormones. Seizure 2015, 28, 12–17. [Google Scholar] [CrossRef] [Green Version]
- Isojarvi, J.I.; Lofgren, E.; Juntunen, K.S.; Pakarinen, A.J.; Paivansalo, M.; Rautakorpi, I.; Tuomivaara, L. Effect of epilepsy and antiepileptic drugs on male reproductive health. Neurology 2004, 62, 247–253. [Google Scholar] [CrossRef]
- Herzog, A.G. Disorders of reproduction in patients with epilepsy: Primary neurological mechanisms. Seizure 2008, 17, 101–110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Talbot, J.A.; Sheldrick, R.; Caswell, H.; Duncan, S. Sexual function in men with epilepsy: How important is testosterone? Neurology 2008, 70, 1346–1352. [Google Scholar] [CrossRef] [PubMed]
- Hamed, S. The effect of epilepsy and antiepileptic drugs on sexual, reproductive and gonadal health of adults with epilepsy. Expert Rev. Clin. Pharmacol. 2016, 9, 807–819. [Google Scholar] [CrossRef] [PubMed]
- Matuszewska, A.; Nowak, B.; Szeląg, A.; Merwid-Ląd, A.; Partyka, A.; Tomkalski, T.; Podhorska-Okołów, M.; Piotrowska, A.; Kwiatkowska, J.; Niżański, W. Long-term stiripentol administration, an anticonvulsant drug, does not impair sperm parameters in rats. Andrologia 2021, 53, e14058. [Google Scholar] [CrossRef] [PubMed]
- Goltzman, D. Functions of vitamin D in bone. Histochem. Cell Biol. 2018, 149, 305–312. [Google Scholar] [CrossRef]
- Watanabe, M.; Maemura, K.; Kanbara, K.; Tamayama, T.; Hayasaki, H. GABA and GABA Receptors in the Central Nervous System and Other Organs. Adv. Virus Res. 2002, 213, 1–47. [Google Scholar] [CrossRef]
- Tamayama, T.; Maemura, K.; Kanbara, K.; Hayasaki, H.; Yabumoto, Y.; Yuasa, M.; Watanabe, M. Expression of GABA(A) and GABA(B) receptors in rat growth plate chondrocytes: Activation of the GABA receptors promotes proliferation of mouse chondrogenic ATDC5 cells. Mol. Cell. Biochem. 2005, 273, 117–126. [Google Scholar] [CrossRef]
- Tamayama, T.; Kanbara, K.; Maemura, K.; Kuno, M.; Watanabe, M. Localization of GABA, GAD65 and GAD67 in Rat Epiphyseal Growth Plate Chondrocytes. Acta Histochem. Cytochem. 2001, 34, 201–206. [Google Scholar] [CrossRef] [Green Version]
- Morgan, E.F.; Unnikrisnan, G.U.; Hussein, A.I. Bone Mechanical Properties in Healthy and Diseased States. Ann. Rev. Biomed. Eng. 2018, 20, 119–143. [Google Scholar] [CrossRef] [PubMed]
- Tran, A.; Tréluyer, J.-M.; Rey, E.; Barbet, J.; Ferracci, G.; D’Athis, P.; Vincent, J.; Pons, G. Protective Effect of Stiripentol on Acetaminophen-Induced Hepatotoxicity in Rat. Toxicol. Appl. Pharmacol. 2001, 170, 145–152. [Google Scholar] [CrossRef]
- Maurizis, J.C.; Rapp, M.; Madelmont, J.C.; Gillardin, J.M.; Lepage, F.; Labarre, P.; Dupuy, J.M.; Veyre, A. Disposition of stiripentol in the pregnant and non-pregnant female rat. Xenobiotica 1993, 23, 1335–1343. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Tang, C.; Rashed, M.; Cui, D.; Tombret, F.; Botte, H.; Lepage, F.; Levy, R.H.; Baillie, T.A. Metabolic chiral inversion of stiripentol in the rat. I. Mechanistic studies. Drug Metab. Dispos. 1994, 22, 544–553. [Google Scholar] [PubMed]
- Nair, A.B.; Jacob, S. A simple practice guide for dose conversion between animals and human. J. Basic Clin. Pharm. 2016, 7, 27–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ozturan, O.; Dogan, R.; Tugrul, S.; Gedik, O.; Sjostrand, A.P.; Yildirim, Y.S. Dose and time dependent ototoxicity of aspartame in rats. Acta Otolaryngol. 2017, 137, 421–425. [Google Scholar] [CrossRef]
- Gámez, R.; Más, R.; Noa, M.; Menéndez, R.; Garcia, H.; González, J.; Pérez, Y.; Goicochea, E. Six-Month Toxicity Study of Oral Administration of D-003 in Sprague Dawley Rats. Drugs R&D 2002, 3, 375–386. [Google Scholar] [CrossRef]
- Shin, J.W.; Kim, H.G.; Park, H.J.; Sung, N.W.; Son, C.G. Safety of the traditional Korean herbal medicine CGX: A 6-month repeated-dose study in rats. J. Ethnopharmacol. 2010, 128, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Bouxsein, M.L.; Boyd, S.K.; Christiansen, B.A.; Guldberg, R.E.; Jepsen, K.J.; Muller, R. Guidelines for assessment of bone microstructure in rodents using micro-computed tomography. J. Bone Miner. Res. 2010, 25, 1468–1486. [Google Scholar] [CrossRef] [PubMed]
- Leppänen, O.; Sievänen, H.; Jokihaara, J.; Pajamäki, I.; Järvinen, T.L. Three-Point Bending of Rat Femur in the Mediolateral Direction: Introduction and Validation of a Novel Biomechanical Testing Protocol. J. Bone Miner. Res. 2006, 21, 1231–1237. [Google Scholar] [CrossRef] [PubMed]


| Serum Parameters | Control Group | STP Group | p Value | |
|---|---|---|---|---|
| Week 8 | PINP (ng/mL) | 3.17 (2.37–3.54) | 2.94 (2.04–3.37) | 0.005 |
| TRAP (U/L) | 0.92 (0.68–0.94) | 0.74 (0.66–0.91) | NS | |
| 25-OH-D (nmol/L) | 5.51 (5.12–5.76) | 6.17 (3.84–6.71) | NS | |
| Week 16 | PINP (ng/mL) * | 2.61 (1.76–2.73) | 1.70 (1.40–1.87) | NS |
| TRAP (U/L) | 0.77 (0.63–0.82) | 0.78 (0.69–0.89) | NS | |
| 25-OH-D (nmol/L) | 6.12 (5.08–6.59) | 5.45 (4.41–5.91) | NS | |
| Total calcium (mg/dL) | 8.00 (7.40–9.10) | 7.40 (7.00–8.90) | NS | |
| Inorganic phosphate (mg/dL) | 3.80 (3.60–4.80) | 4.40 (3.70–5.20) | NS | |
| Week 24 | PINP (ng/mL) | 0.84 (0.78–1.00) | 0.78 (0.69–0.99) | NS |
| TRAP (U/L) | 0.69 (0.64–0.84) | 0.82 (0.66–0.90) | NS | |
| 25-OH-D (nmol/L) | 5.66 (5.18–5.97) | 5.25 (4.47–6.44) | NS | |
| 1,25-dihydroxyvitamin D3 (nmol/L) * | 1.42 (1.37–1.69) | 1.33 (1.25–1.46) | 0.04 | |
| Parathormone (pg/mL) | 536.1 (417.0–754.2) | 464.7 (340.4–570.0) | NS | |
| IGF-1 (ng/mL) | 1.58 (1.29–2.38) | 1.35 (1.18–1.47) | NS | |
| Total calcium (mg/dL) | 9.65 (9.35–9.95) | 9.70 (9.50–10.05) | NS | |
| Inorganic phosphate (mg/dL) * | 4.40 (3.85–5.15) | 5.80 (4.95–6.45) | 0.015 | |
| Creatinine (mg/dL) | 0.29 (0.27–0.31) | 0.30 (0.28–0.32) | NS | |
| NGAL (ng/mL) * | 0.27 (0.26–0.29) | 0.30 (0.27–0.31) | 0.03 | |
| Aspartate aminotransferase (U/L) | 189 (139.5–292) | 198 (139.5–263.5) | NS | |
| Alanine aminotransferase (U/L) | 58 (48–70.5) | 49 (39.5–63.5) | NS |
| Bone Macrometric Parameters | Control Group | STP Group | p Value |
|---|---|---|---|
| Tibial index (-) | 0.0032 (0.0029–0.0033) | 0.0032 (0.0029–0.0033) | NS |
| Tibial weight (g) | 0.964 (0.930–1.044) | 0.9210 (0.895–0.960) | NS |
| Tibia length (mm) | 43.33 (42.62–43.77) | 42.57 (42.50–43.05) | NS |
| Mid-tibial diameter (mm) | 3.735 (3.625–3.850) | 3.720 (3.640–3.945) | NS |
| Femur index (-) | 0.0021 (0.0021–0.0023) | 0.0021 (0.0020–0.0023) | NS |
| Femur weight (g) | 1.372 (1.334–1.431) | 1.322 (1.270–1.406) | NS |
| Femur length (mm) | 39.77 (39.38–40.37) | 39.70 (39.21–40.13) | NS |
| Mid-femoral diameter (mm) | 4.815 (4.710–4.915) | 4.680 (4.640–4.970) | NS |
| Bone Mineral Density | Control Group | STP Group | p Value |
|---|---|---|---|
| Tibial BMD (g/cm2) | 0.240 (0.231–0.243) | 0.234 (0.232–0.241) | NS |
| Femoral BMD (g/cm2) | 0.291 (0.279–0.294) | 0.282 (0.275–0.290) | NS |
| L1–L4 BMD (g/cm2) | 0.340 (0.334–0.360) | 0.324 (0.320–0.342) | NS |
| Bone Parameters | Control Group | STP Group | p Value | ||
|---|---|---|---|---|---|
| Tibia | Cancellous bone | BV/TV (%) * | 9.28 (8.63–10.20) | 8.02 (7.70–8.60) | 0.03 |
| BS/TV (mm3/mm2) | 4.62 (4.25–5.54) | 4.37 (4.17–5.42) | NS | ||
| Tb.Th (mm) * | 0.078 (0.07–0.08) | 0.075 (0.07–0.08) | 0.03 | ||
| Tb.N (1/mm) | 1.20 (1.13–1.39) | 1.08 (1.03–1.47) | NS | ||
| Tb.Sp (mm) | 0.70 (0.63–1.10) | 0.64 (0.46–0.80) | NS | ||
| Tb.Pf (1/mm) * | 19.76 (18.13–20.27) | 21.26 (20.21–23.37) | 0.02 | ||
| SMI (-) * | 2.30 (2.23–2.32) | 2.40 (2.32–2.59) | 0.006 | ||
| DA (-) | 1.47 (1.44–1.62) | 1.55 (1.32–1.85) | NS | ||
| Conn.D (1/mm3) | 33.16 (26.38–37.31) | 23.67 (17.84–37.33) | NS | ||
| Po.tot (%) | 90.72 (89.81–91.37) | 91.75 (89.74–92.47) | NS | ||
| Cortical bone | Cr.Th (mm) | 0.65 (0.63–0.67) | 0.65 (0.62–0.66) | NS | |
| Tt.Ar (mm2) | 52.49 (50.37–53.79) | 51.28 (50.42–54.72) | NS | ||
| Ct.Ar (mm2) | 65.79 (63.57–67.04) | 64.55 (61.95–67.72) | NS | ||
| Tt.Ar/Ct.Ar (%) | 80.40 (78.92–81.21) | 79.56 (78.59–81.77) | NS | ||
| Femur | Cancellous bone | BV/TV (%) | 14.47 (12.95–16.76) | 12.63 (11.58–18.95) | NS |
| BS/TV (mm3/mm2) | 6.55 (6.14–7.93) | 6.04 (5.65–8.56) | NS | ||
| Tb.Th (mm) | 0.079 (0.77–0.085) | 0.077 (0.076–0.081) | NS | ||
| Tb.N (1/mm) | 1.77 (1.67–2.11) | 1.61 (1.51–2.33) | NS | ||
| Tb.Sp (mm) | 0.60 (0.49–0.86) | 0.56 (0.45–0.78) | NS | ||
| Tb.Pf (1/mm) | 15.35 (13.94 - 18.91) | 16.64 (15.46–18.59) | NS | ||
| SMI (-) | 1.92 (1.82–2.11) | 2.03 (1.93–2.19) | NS | ||
| DA (-) | 1.25 (1.19–1.38) | 1.32 (1.26–1.44) | NS | ||
| Conn.D (1/mm3) | 65.20 (59.08–84.63) | 54.02 (51.29–86.07) | NS | ||
| Po.tot (%) | 85.53 (85.19–89.39) | 87.37 (85.27–90.20) | NS | ||
| Cortical bone | Cr.Th (mm) | 0.75 (0.74–0.78) | 0.76 (0.72–0.80) | NS | |
| Tt.Ar (mm2) | 52.77 (52.03–54.57) | 50.97 (49.92–54.05) | NS | ||
| Ct.Ar (mm2) | 62.00 (60.41–63.56) | 60.41 (58.29–63.57) | NS | ||
| Tt.Ar/Ct.Ar (%) | 86.23 (85,58–86.76) | 85.55 (84.74–86.38) | NS | ||
| L3 vertebra | Cancellous bone | BV/TV (%) | 19.08 (17.59–21.82) | 17.79 (16.57–22.69) | NS |
| BS/TV (mm3/mm2) | 8.67 (7.88–9.69) | 8.42 (7.67–10.73) | NS | ||
| Tb.Th (mm) | 0.078 (0.077–0.079) | 0.076 (0.075–0.081) | NS | ||
| Tb.N (1/mm) | 2.44 (2.22–2.82) | 2.36 (2.15–3.03) | NS | ||
| Tb.Sp (mm) | 0.27 (0.26–0.33) | 0.27 (0.25–0.37) | NS | ||
| Tb.Pf (1/mm) | 12.42 (11.95–15.39) | 13.47 (12.88–15.86) | NS | ||
| SMI (-) | 1.65 (1.67–1.95) | 1.73 (1.70–1.97) | NS | ||
| DA (-) | 1.44 (1.35–1.61) | 1.41 (1.34–1.52) | NS | ||
| Conn.D (1/mm3) | 74.70 (63.79–108.88) | 70.87 (61.4–118.15) | NS | ||
| Po.tot (%) | 80.92 (80.21–86.03) | 82.21 (80.13–86.07) | NS |
| Bone Mechanical Properties | Control Group | STP Group | p Value | |
|---|---|---|---|---|
| Tibia | Young’s modulus (GPa) | 11.12 (10.02–12.67) | 9.51 (8.67–11.24) | 0.044 |
| Bending strength (MPa) | 251.66 (237.96–273.53) | 244.33 (232.60–248.92) | NS | |
| Bending stiffness (Nm2) | 0.0431 (0.0401–0.0531) | 0.0393 (0.0333–0.0404) | 0.024 | |
| Femur | Young’s modulus (GPa) | 6.13 (5.67–8.78) | 6.31 (5.72–6.89) | NS |
| Bending strength (MPa) | 166.76 (157.44–184.17) | 162.07 (158.06–183.15) | NS | |
| Bending stiffness (Nm2) | 0.06 (0.05–0.08) | 0.05 (0.05–0.07) | NS |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matuszewska, A.; Nowak, B.; Nikodem, A.; Merwid-Ląd, A.; Wiatrak, B.; Tomkalski, T.; Jędrzejuk, D.; Szeląg, E.; Sozański, T.; Danielewski, M.; et al. Antiepileptic Stiripentol May Influence Bones. Int. J. Mol. Sci. 2021, 22, 7162. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22137162
Matuszewska A, Nowak B, Nikodem A, Merwid-Ląd A, Wiatrak B, Tomkalski T, Jędrzejuk D, Szeląg E, Sozański T, Danielewski M, et al. Antiepileptic Stiripentol May Influence Bones. International Journal of Molecular Sciences. 2021; 22(13):7162. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22137162
Chicago/Turabian StyleMatuszewska, Agnieszka, Beata Nowak, Anna Nikodem, Anna Merwid-Ląd, Benita Wiatrak, Tomasz Tomkalski, Diana Jędrzejuk, Ewa Szeląg, Tomasz Sozański, Maciej Danielewski, and et al. 2021. "Antiepileptic Stiripentol May Influence Bones" International Journal of Molecular Sciences 22, no. 13: 7162. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22137162







