Mediators of Metabolism: An Unconventional Role for NOD1 and NOD2
Abstract
:1. Introduction
2. Contribution of NOD1 and NOD2 to Metabolic Diseases
2.1. Links between Human Metabolic Diseases and NOD1 or NOD2
2.2. High Fat Diet Consumption Upregulates NOD1 and NOD2 Signaling Pathways in Mice
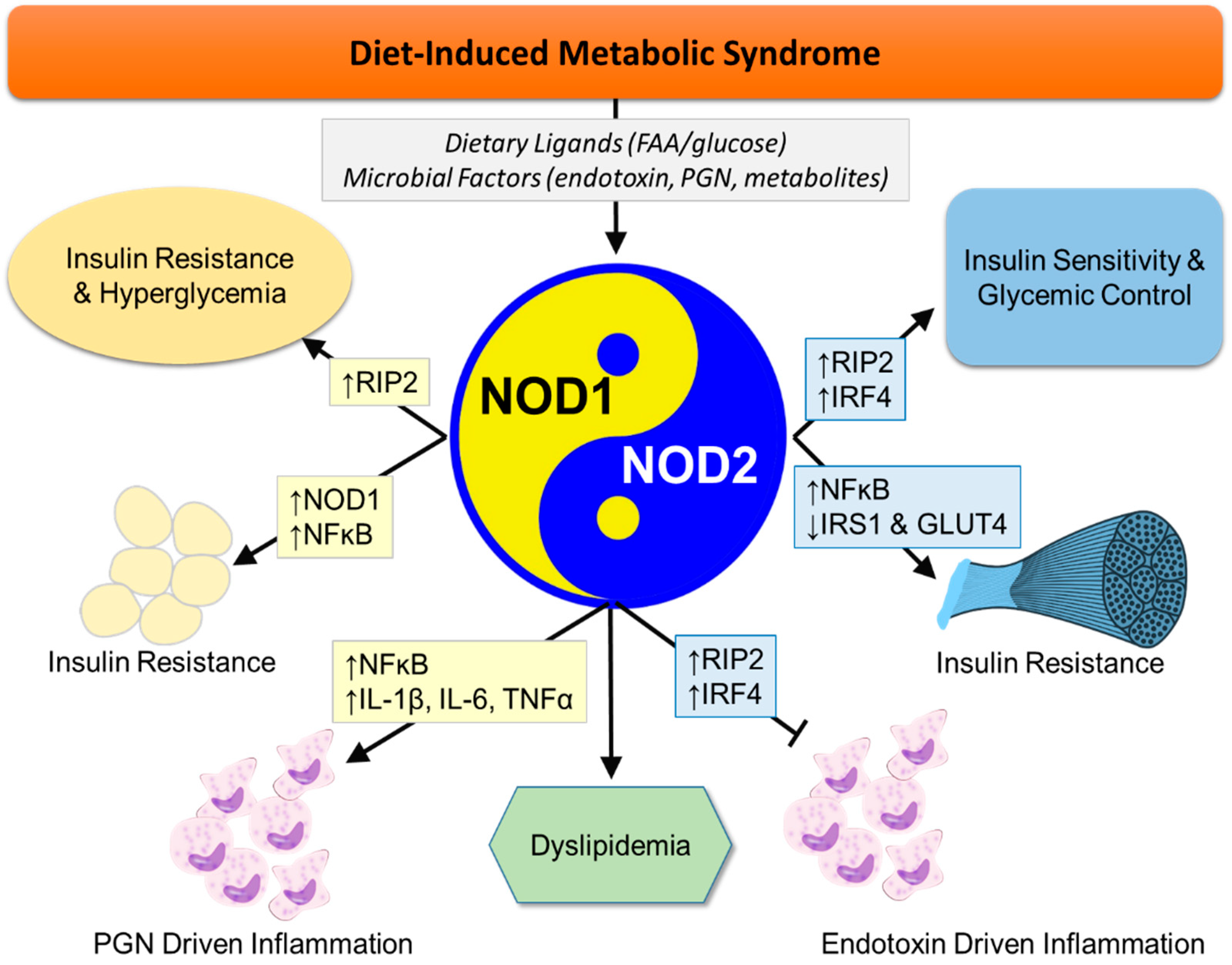
2.3. Opposing Roles of NOD1 and NOD2 in Metabolic Disease
3. NOD1 and NOD2 as Modulators of Cellular Responses to Glucose and Fatty Acids
3.1. Roles of NOD1 and NOD2 in Hyperglycemia and Insulin Resistance
3.2. Fatty Acids as Ligands for NOD1 and NOD2
4. NOD1 and NOD2 as Mediators of Cellular Metabolic Stress
4.1. Endoplasmic Reticulum Stress
4.2. Calcium Signaling
4.3. Reactive Oxygen Species Generation and Mitochondrial Dysfunction
5. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Caruso, R.; Warner, N.; Inohara, N.; Nunez, G. NOD1 and NOD2: Signaling, host defense, and inflammatory disease. Immunity 2014, 41, 898–908. [Google Scholar] [CrossRef] [Green Version]
- Mukherjee, T.; Hovingh, E.S.; Foerster, E.G.; Abdel-Nour, M.; Philpott, D.J.; Girardin, S.E. NOD1 and NOD2 in inflammation, immunity and disease. Arch. Biochem. Biophys. 2019, 670, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Hotamisligil, G.S. Inflammation and metabolic disorders. Nature 2006, 444, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Cavallari, J.F.; Pokrajac, N.T.; Zlitni, S.; Foley, K.P.; Henriksbo, B.D.; Schertzer, J.D. NOD2 in hepatocytes engages a liver-gut axis to protect against steatosis, fibrosis, and gut dysbiosis during fatty liver disease in mice. Am. J. Physiol. Endocrinol. Metab. 2020, 319, E305–E314. [Google Scholar] [CrossRef]
- Cuda, C.; Badawi, A.; Karmali, M.; El-Sohemy, A. Effects of polymorphisms in nucleotide-binding oligomerization domains 1 and 2 on biomarkers of the metabolic syndrome and type II diabetes. Genes Nutr. 2012, 7, 427–435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lappas, M. NOD1 expression is increased in the adipose tissue of women with gestational diabetes. J. Endocrinol. 2014, 222, 99–112. [Google Scholar] [CrossRef] [Green Version]
- Schertzer, J.D.; Tamrakar, A.K.; Magalhaes, J.G.; Pereira, S.; Bilan, P.J.; Fullerton, M.D.; Liu, Z.; Steinberg, G.R.; Giacca, A.; Philpott, D.J.; et al. NOD1 activators link innate immunity to insulin resistance. Diabetes 2011, 60, 2206–2215. [Google Scholar] [CrossRef]
- Shiny, A.; Regin, B.; Balachandar, V.; Gokulakrishnan, K.; Mohan, V.; Babu, S.; Balasubramanyam, M. Convergence of innate immunity and insulin resistance as evidenced by increased nucleotide oligomerization domain (NOD) expression and signaling in monocytes from patients with type 2 diabetes. Cytokine 2013, 64, 564–570. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Y.J.; Liu, C.; Li, C.L.; Song, Y.L.; Tang, Y.S.; Zhou, H.; Li, A.; Li, Y.; Weng, Y.; Zheng, F.P. Increased NOD1, but not NOD2, activity in subcutaneous adipose tissue from patients with metabolic syndrome. Obesity 2015, 23, 1394–1400. [Google Scholar] [CrossRef]
- Ruiz, E.; Penrose, H.M.; Heller, S.; Nakhoul, H.; Baddoo, M.; Flemington, E.F.; Kandil, E.; Savkovic, S.D. Bacterial TLR4 and NOD2 signaling linked to reduced mitochondrial energy function in active inflammatory bowel disease. Gut Microbes 2020, 11, 350–363. [Google Scholar] [CrossRef]
- Zhou, Y.J.; Tang, Y.S.; Song, Y.L.; Li, A.; Zhou, H.; Li, Y. Saturated fatty acid induces insulin resistance partially through nucleotide-binding oligomerization domain 1 signaling pathway in adipocytes. Chin. Med. Sci. J. 2013, 28, 211–217. [Google Scholar] [CrossRef]
- Fan, Y.H.; Roy, S.; Mukhopadhyay, R.; Kapoor, A.; Duggal, P.; Wojcik, G.L.; Pass, R.F.; Arav-Boger, R. Role of nucleotide-binding oligomerization domain 1 (NOD1) and its variants in human cytomegalovirus control in vitro and in vivo. Proc. Natl. Acad. Sci. USA 2016, 113, E7818–E7827. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ozbayer, C.; Kurt, H.; Kebapci, M.N.; Gunes, H.V.; Colak, E.; Degirmenci, I. Effects of genetic variations in the genes encoding NOD1 and NOD2 on type 2 diabetes mellitus and insulin resistance. J. Clin. Pharm. Ther. 2017, 42, 98–102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Winzell, M.S.; Ahren, B. The high-fat diet-fed mouse: A model for studying mechanisms and treatment of impaired glucose tolerance and type 2 diabetes. Diabetes 2004, 53 (Suppl. 3), S215–S219. [Google Scholar] [CrossRef] [Green Version]
- Martinez-Medina, M.; Denizot, J.; Dreux, N.; Robin, F.; Billard, E.; Bonnet, R.; Darfeuille-Michaud, A.; Barnich, N. Western diet induces dysbiosis with increased E coli in CEABAC10 mice, alters host barrier function favouring AIEC colonisation. Gut 2014, 63, 116–124. [Google Scholar] [CrossRef]
- Zhao, L.; Hu, P.; Zhou, Y.; Purohit, J.; Hwang, D. NOD1 activation induces proinflammatory gene expression and insulin resistance in 3T3-L1 adipocytes. Am. J. Physiol. Endocrinol. Metab. 2011, 301, E587–E598. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Y.J.; Zhou, H.; Li, Y.; Song, Y.L. NOD1 activation induces innate immune responses and insulin resistance in human adipocytes. Diabetes Metab. 2012, 38, 538–543. [Google Scholar] [CrossRef]
- Kim, M.S.; Choi, M.S.; Han, S.N. High fat diet-induced obesity leads to proinflammatory response associated with higher expression of NOD2 protein. Nutr. Res. Pract. 2011, 5, 219–223. [Google Scholar] [CrossRef]
- Chan, K.L.; Tam, T.H.; Boroumand, P.; Prescott, D.; Costford, S.R.; Escalante, N.K.; Fine, N.; Tu, Y.; Robertson, S.J.; Prabaharan, D.; et al. Circulating NOD1 Activators and Hematopoietic NOD1 Contribute to Metabolic Inflammation and Insulin Resistance. Cell Rep. 2017, 18, 2415–2426. [Google Scholar] [CrossRef] [Green Version]
- Bae, J.; Ricciardi, C.J.; Esposito, D.; Komarnytsky, S.; Hu, P.; Curry, B.J.; Brown, P.L.; Gao, Z.; Biggerstaff, J.P.; Chen, J.; et al. Activation of pattern recognition receptors in brown adipocytes induces inflammation and suppresses uncoupling protein 1 expression and mitochondrial respiration. Am. J. Physiol Cell Physiol. 2014, 306, C918–C930. [Google Scholar] [CrossRef] [Green Version]
- Mendez, J.M.; Kolora, L.D.; Lemon, J.S.; Dupree, S.L.; Keestra-Gounder, A.M. Activation of the Endoplasmic Reticulum Stress Response Impacts the NOD1 Signaling Pathway. Infect. Immun. 2019, 87. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cavallari, J.F.; Barra, N.G.; Foley, K.P.; Lee, A.; Duggan, B.M.; Henriksbo, B.D.; Anhe, F.F.; Ashkar, A.A.; Schertzer, J.D. Postbiotics for NOD2 require nonhematopoietic RIPK2 to improve blood glucose and metabolic inflammation in mice. Am. J. Physiol. Endocrinol. Metab. 2020, 318, E579–E585. [Google Scholar] [CrossRef] [PubMed]
- Cavallari, J.F.; Fullerton, M.D.; Duggan, B.M.; Foley, K.P.; Denou, E.; Smith, B.K.; Desjardins, E.M.; Henriksbo, B.D.; Kim, K.J.; Tuinema, B.R.; et al. Muramyl dipeptide-based postbiotics mitigate obesity-induced insulin resistance via IRF4. Cell Metab. 2017, 25, 1063–1074.e3. [Google Scholar] [CrossRef] [PubMed]
- Tamrakar, A.K.; Schertzer, J.D.; Chiu, T.T.; Foley, K.P.; Bilan, P.J.; Philpott, D.J.; Klip, A. NOD2 activation induces muscle cell-autonomous innate immune responses and insulin resistance. Endocrinology 2010, 151, 5624–5637. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maurya, C.K.; Arha, D.; Rai, A.K.; Kumar, S.K.; Pandey, J.; Avisetti, D.R.; Kalivendi, S.V.; Klip, A.; Tamrakar, A.K. NOD2 activation induces oxidative stress contributing to mitochondrial dysfunction and insulin resistance in skeletal muscle cells. Free Radic. Biol. Med. 2015, 89, 158–169. [Google Scholar] [CrossRef]
- Levy, A.; Stedman, A.; Deutsch, E.; Donnadieu, F.; Virgin, H.W.; Sansonetti, P.J.; Nigro, G. Innate immune receptor NOD2 mediates LGR5(+) intestinal stem cell protection against ROS cytotoxicity via mitophagy stimulation. Proc. Natl. Acad. Sci. USA 2020, 117, 1994–2003. [Google Scholar] [CrossRef] [Green Version]
- Chapman, T.P.; Corridoni, D.; Shiraishi, S.; Pandey, S.; Aulicino, A.; Wigfield, S.; do Carmo Costa, M.; Thezenas, M.L.; Paulson, H.; Fischer, R.; et al. Ataxin-3 Links NOD2 and TLR2 Mediated Innate Immune Sensing and Metabolism in Myeloid Cells. Front. Immunol. 2019, 10, 1495. [Google Scholar] [CrossRef] [Green Version]
- Huang, W.; Gou, F.; Long, Y.; Li, Y.; Feng, H.; Zhang, Q.; Gao, C.; Chen, G.; Xu, Y. High Glucose and Lipopolysaccharide Activate NOD1- RICK-NF-kappaB Inflammatory Signaling in Mesangial Cells. Exp. Clin. Endocrinol. Diabetes 2016, 124, 512–517. [Google Scholar] [CrossRef]
- Du, P.; Fan, B.; Han, H.; Zhen, J.; Shang, J.; Wang, X.; Li, X.; Shi, W.; Tang, W.; Bao, C.; et al. NOD2 promotes renal injury by exacerbating inflammation and podocyte insulin resistance in diabetic nephropathy. Kidney Int. 2013, 84, 265–276. [Google Scholar] [CrossRef] [Green Version]
- Zhao, L.; Kwon, M.J.; Huang, S.; Lee, J.Y.; Fukase, K.; Inohara, N.; Hwang, D.H. Differential modulation of Nods signaling pathways by fatty acids in human colonic epithelial HCT116 cells. J. Biol. Chem. 2007, 282, 11618–11628. [Google Scholar] [CrossRef] [Green Version]
- Keestra-Gounder, A.M.; Byndloss, M.X.; Seyffert, N.; Young, B.M.; Chavez-Arroyo, A.; Tsai, A.Y.; Cevallos, S.A.; Winter, M.G.; Pham, O.H.; Tiffany, C.R.; et al. NOD1 and NOD2 signalling links ER stress with inflammation. Nature 2016, 532, 394–397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, C.; Hedl, M.; Ranjan, K.; Abraham, C. LACC1 Required for NOD2-Induced, ER Stress-Mediated Innate Immune Outcomes in Human Macrophages and LACC1 Risk Variants Modulate These Outcomes. Cell Rep. 2019, 29, 4525–4539.e4524. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murugina, N.E.; Budikhina, A.S.; Dagil, Y.A.; Maximchik, P.V.; Balyasova, L.S.; Murugin, V.V.; Melnikov, M.V.; Sharova, V.S.; Nikolaeva, A.M.; Chkadua, G.Z.; et al. Glycolytic reprogramming of macrophages activated by NOD1 and TLR4 agonists: No association with proinflammatory cytokine production in normoxia. J. Biol. Chem. 2020, 295, 3099–3114. [Google Scholar] [CrossRef] [PubMed]
- Molinaro, R.; Mukherjee, T.; Flick, R.; Philpott, D.J.; Girardin, S.E. Trace levels of peptidoglycan in serum underlie the NOD-dependent cytokine response to endoplasmic reticulum stress. J. Biol. Chem. 2019, 294, 9007–9015. [Google Scholar] [CrossRef]
- Kwon, M.Y.; Hwang, N.; Lee, S.J.; Chung, S.W. Nucleotide-binding oligomerization domain protein 2 attenuates ER stress-induced cell death in vascular smooth muscle cells. BMB Rep. 2019, 52, 665–670. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, X.; Zheng, G.; Shan, Q.; Lu, J.; Fan, S.; Sun, C.; Wu, D.; Zhang, C.; Su, W.; et al. Troxerutin attenuates enhancement of hepatic gluconeogenesis by inhibiting nod activation-mediated inflammation in high-fat diet-treated mice. Int. J. Mol. Sci. 2016, 18, 31. [Google Scholar] [CrossRef] [Green Version]
- Ozcan, U.; Yilmaz, E.; Ozcan, L.; Furuhashi, M.; Vaillancourt, E.; Smith, R.O.; Gorgun, C.Z.; Hotamisligil, G.S. Chemical chaperones reduce ER stress and restore glucose homeostasis in a mouse model of type 2 diabetes. Science 2006, 313, 1137–1140. [Google Scholar] [CrossRef] [Green Version]
- Amar, J.; Chabo, C.; Waget, A.; Klopp, P.; Vachoux, C.; Bermudez-Humaran, L.G.; Smirnova, N.; Berge, M.; Sulpice, T.; Lahtinen, S.; et al. Intestinal mucosal adherence and translocation of commensal bacteria at the early onset of type 2 diabetes: Molecular mechanisms and probiotic treatment. EMBO Mol. Med. 2011, 3, 559–572. [Google Scholar] [CrossRef]
- Carlos, D.; Perez, M.M.; Leite, J.A.; Rocha, F.A.; Martins, L.M.S.; Pereira, C.A.; Fraga-Silva, T.F.C.; Pucci, T.A.; Ramos, S.G.; Camara, N.O.S.; et al. NOD2 Deficiency Promotes Intestinal CD4+ T Lymphocyte Imbalance, Metainflammation, and Aggravates Type 2 Diabetes in Murine Model. Front. Immunol. 2020, 11, 1265. [Google Scholar] [CrossRef]
- Denou, E.; Lolmede, K.; Garidou, L.; Pomie, C.; Chabo, C.; Lau, T.C.; Fullerton, M.D.; Nigro, G.; Zakaroff-Girard, A.; Luche, E.; et al. Defective NOD2 peptidoglycan sensing promotes diet-induced inflammation, dysbiosis, and insulin resistance. EMBO Mol. Med. 2015, 7, 259–274. [Google Scholar] [CrossRef]
- Rodriguez-Nunez, I.; Caluag, T.; Kirby, K.; Rudick, C.N.; Dziarski, R.; Gupta, D. Nod2 and Nod2-regulated microbiota protect BALB/c mice from diet-induced obesity and metabolic dysfunction. Sci. Rep. 2017, 7, 548. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eguchi, J.; Kong, X.; Tenta, M.; Wang, X.; Kang, S.; Rosen, E.D. Interferon regulatory factor 4 regulates obesity-induced inflammation through regulation of adipose tissue macrophage polarization. Diabetes 2013, 62, 3394–3403. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watanabe, T.; Asano, N.; Murray, P.J.; Ozato, K.; Tailor, P.; Fuss, I.J.; Kitani, A.; Strober, W. Muramyl dipeptide activation of nucleotide-binding oligomerization domain 2 protects mice from experimental colitis. J. Clin. Invest. 2008, 118, 545–559. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagpal, R.; Newman, T.M.; Wang, S.; Jain, S.; Lovato, J.F.; Yadav, H. Obesity-Linked gut microbiome dysbiosis associated with derangements in gut permeability and intestinal cellular homeostasis independent of diet. J. Diabetes Res. 2018, 2018, 3462092. [Google Scholar] [CrossRef]
- de Kort, S.; Keszthelyi, D.; Masclee, A.A. Leaky gut and diabetes mellitus: What is the link? Obes. Rev. 2011, 12, 449–458. [Google Scholar] [CrossRef]
- Thaiss, C.A.; Levy, M.; Grosheva, I.; Zheng, D.; Soffer, E.; Blacher, E.; Braverman, S.; Tengeler, A.C.; Barak, O.; Elazar, M.; et al. Hyperglycemia drives intestinal barrier dysfunction and risk for enteric infection. Science 2018, 359, 1376–1383. [Google Scholar] [CrossRef] [Green Version]
- DeFronzo, R.A.; Tripathy, D. Skeletal Muscle Insulin Resistance Is the Primary Defect in Type 2 Diabetes. J. Diabetes Care 2009, 32, S157–S163. [Google Scholar] [CrossRef] [Green Version]
- Cascio, G.; Schiera, G.; Di Liegro, I. Dietary fatty acids in metabolic syndrome, diabetes and cardiovascular diseases. Curr Diabetes Rev. 2012, 8, 2–17. [Google Scholar] [CrossRef]
- Chi, W.; Dao, D.; Lau, T.C.; Henriksbo, B.D.; Cavallari, J.F.; Foley, K.P.; Schertzer, J.D. Bacterial peptidoglycan stimulates adipocyte lipolysis via NOD1. PLoS ONE 2014, 9, e97675. [Google Scholar] [CrossRef]
- Purohit, J.S.; Hu, P.; Chen, G.; Whelan, J.; Moustaid-Moussa, N.; Zhao, L. Activation of nucleotide oligomerization domain containing protein 1 induces lipolysis through NF-κB and the lipolytic PKA activation in 3T3-L1 adipocytes. Biochem. Cell Biol. Biochim. Biol. Cell. 2013, 91, 428–434. [Google Scholar] [CrossRef]
- Morigny, P.; Houssier, M.; Mouisel, E.; Langin, D. Adipocyte lipolysis and insulin resistance. Biochimie 2016, 125, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Oakes, S.A.; Papa, F.R. The role of endoplasmic reticulum stress in human pathology. Annu. Rev. Pathol. 2015, 10, 173–194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hetz, C. The unfolded protein response: Controlling cell fate decisions under ER stress and beyond. Nat. Rev. Mol. Cell Biol. 2012, 13, 89–102. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Han, K.; Tilve, S.; Wu, K.; Geller, H.M.; Sack, M.N. Parkin targets NOD2 to regulate astrocyte endoplasmic reticulum stress and inflammation. Glia 2018, 66, 2427–2437. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Zhang, W.; Xu, C.; Zhang, S.; Zhao, J.; Pan, Q.; Wang, Z. Nucleotide-binding oligomerization domain protein 1 enhances oxygen-glucose deprivation and reperfusion injury in cortical neurons via activation of endoplasmic reticulum stress-mediated autophagy. Exp. Mol. Pathol. 2020, 117, 104525. [Google Scholar] [CrossRef] [PubMed]
- Wellen, K.E.; Thompson, C.B. Cellular metabolic stress: Considering how cells respond to nutrient excess. Mol. Cell 2010, 40, 323–332. [Google Scholar] [CrossRef] [Green Version]
- GBD 2015 Obesity Collaborators; Afshin, A.; Forouzanfar, M.H.; Reitsma, M.B.; Sur, P.; Estep, K.; Lee, A.; Marczak, L.; Mokdad, A.H.; Moradi-Lakeh, M.; et al. Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N. Engl. J. Med. 2017, 377, 13–27. [Google Scholar] [CrossRef]
- Waters, H.; Graf, M. America’s Obesity Crisis: Economic Costs of Excess Weight; Milken Institute: Santa Monica, CA, USA, 2018. [Google Scholar]
- Le Roux, C.W. COVID-19 alters thinking and management in metabolic diseases. Nat. Rev. Endocrinol. 2020. [Google Scholar] [CrossRef]
- Costa, F.R.; Francozo, M.C.; de Oliveira, G.G.; Ignacio, A.; Castoldi, A.; Zamboni, D.S.; Ramos, S.G.; Camara, N.O.; de Zoete, M.R.; Palm, N.W.; et al. Gut microbiota translocation to the pancreatic lymph nodes triggers NOD2 activation and contributes to T1D onset. J. Exp. Med. 2016, 213, 1223–1239. [Google Scholar] [CrossRef]
| Ligand | Target | NOD1 Action | NOD2 Action |
|---|---|---|---|
| Microbial | |||
| iE-DAP | Adipocytes [16,17,20] | ↑ pro-inflammatory signaling ↑ insulin resistance ↓ glycoysis ↓ OxPhos uncoupling | |
| Epithelial Cells [21] | ↑ pro-inflammatory signaling | ||
| FK156 | Mice on HFD [7,22] | ↑ insulin resistance ↑ glucose intolerance | |
| MDP | Mice on HFD [22,23] | ↑ insulin sensitivity, ↑ glucose tolerance | |
| Skeletal Muscle Cells [24,25] | ↑ insulin resistance ↓ IRS-1 activation, ↓ glucose uptake ↑ mtROS | ||
| Intestinal Stem Cells [26] | ↓ mtROS | ||
| Dendritic Cells [27] | ↑ mtROS and mitochondrial respiration | ||
| Dietary | |||
| Glucose | Mesangial Cells [28] | ↑ pro-inflammatory signaling | |
| Podocytes [29] | ↑ pro-inflammatory signaling | ||
| Saturated Fatty Acids | Adipocytes [11] Fibroblasts [12] Intestinal Epithelial Cells [30] | ↑ pro-inflammatory signaling ↓ insulin-stimulated glucose uptake | ↑ pro-inflammatory signaling |
| Unsaturated Fatty Acids | Intestinal Epithelial Cells [30] | ↓ pro-inflammatory signaling | ↓ pro-inflammatory signaling ↓ NOD2 oligomerization |
| Endoplasmic Stress Agonists | |||
| Thapsigargin | Mice [31] | ↑ IRE1α activation ↑ IL-1β, IL-6, TNFα, MCP-1 | ↑ IRE1α activation ↑ IL-1β, IL-6, TNFα, MCP-1 |
| Macrophages * [31,32,33] | ↑ IRE1α activation * ↑ IL-1β, IL-6, TNFα, MCP-1 | ↑ IRE1α activation * ↑ IL-1β, IL-6, TNFα, MCP-1 | |
| Intestinal Epithelial Cells [34] | ↑ CXCL1, IL-8, CCL20 | ↑ CXCL1, IL-8, CCL20 | |
| Tunicamycin | VSMC [35] | ↑ IRE1α activation ↓ cell death | |
| Endoplasmic Stress Antagonists | |||
| Troxerutin | Mice on HFD [36] | ↓ NOD1 expression ↓ pro-inflammatory signaling | ↓ NOD2 expression ↓ pro-inflammatory signaling |
| TUDCA | Mice [31,37] | ↓ IRE1α activation | ↓ IRE1α activation |
| BMDM [31,37] | ↓ IRE1α activation | ↓ IRE1α activation | |
| PERK Inhibitor GSK2656157 | Epithelial Cells [21] | ↓ pro-inflammatory signaling | |
| Calcium Flux Agonists | |||
| A23187 | Intestinal Epithelial Cells [34] | ↑ CXCL1, IL-8, CCL20 | ↑ CXCL1, IL-8, CCL20 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zangara, M.T.; Johnston, I.; Johnson, E.E.; McDonald, C. Mediators of Metabolism: An Unconventional Role for NOD1 and NOD2. Int. J. Mol. Sci. 2021, 22, 1156. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22031156
Zangara MT, Johnston I, Johnson EE, McDonald C. Mediators of Metabolism: An Unconventional Role for NOD1 and NOD2. International Journal of Molecular Sciences. 2021; 22(3):1156. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22031156
Chicago/Turabian StyleZangara, Megan T., Isabel Johnston, Erin E. Johnson, and Christine McDonald. 2021. "Mediators of Metabolism: An Unconventional Role for NOD1 and NOD2" International Journal of Molecular Sciences 22, no. 3: 1156. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22031156





