Effects of Dietary Protein and Fat Content on Intrahepatocellular and Intramyocellular Lipids during a 6-Day Hypercaloric, High Sucrose Diet: A Randomized Controlled Trial in Normal Weight Healthy Subjects
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
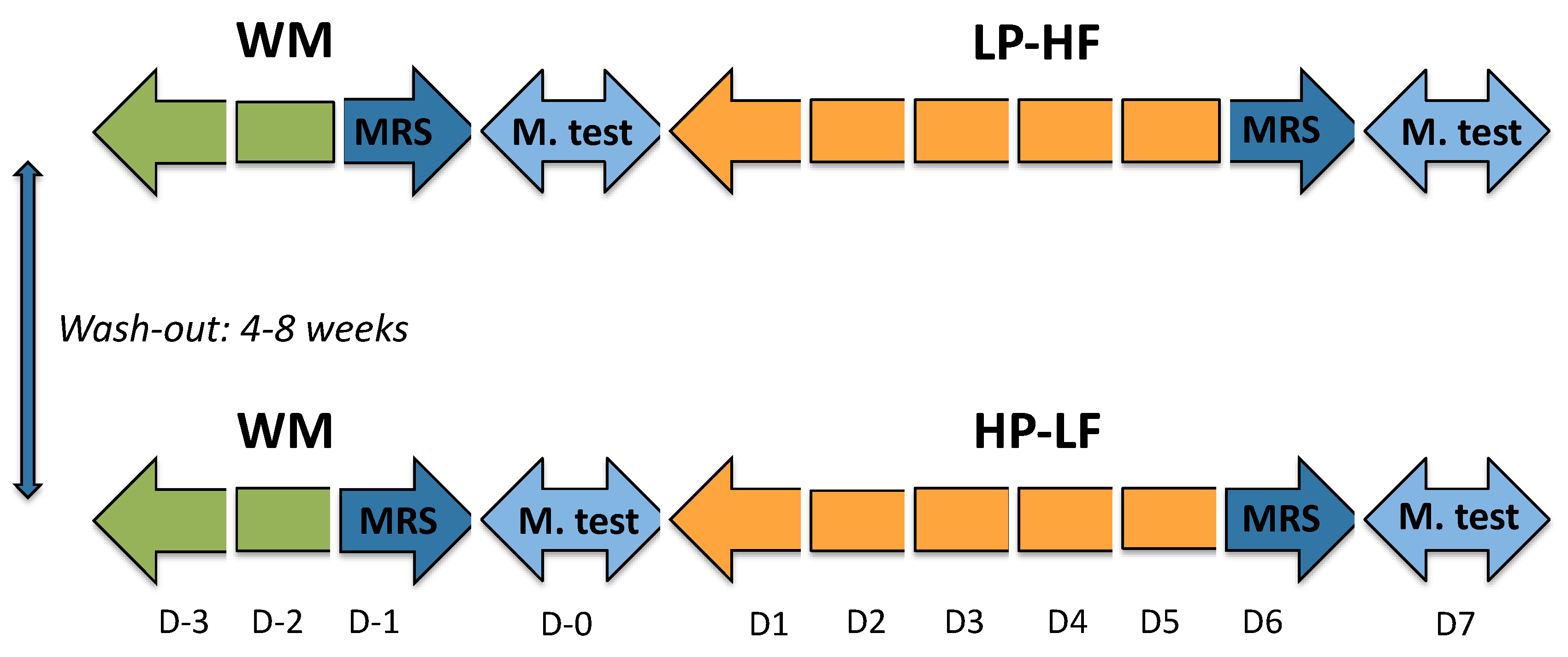
2.2. Experimental Protocol
2.3. Dietary Interventions
2.4. Measurements of IHCL and IMCL
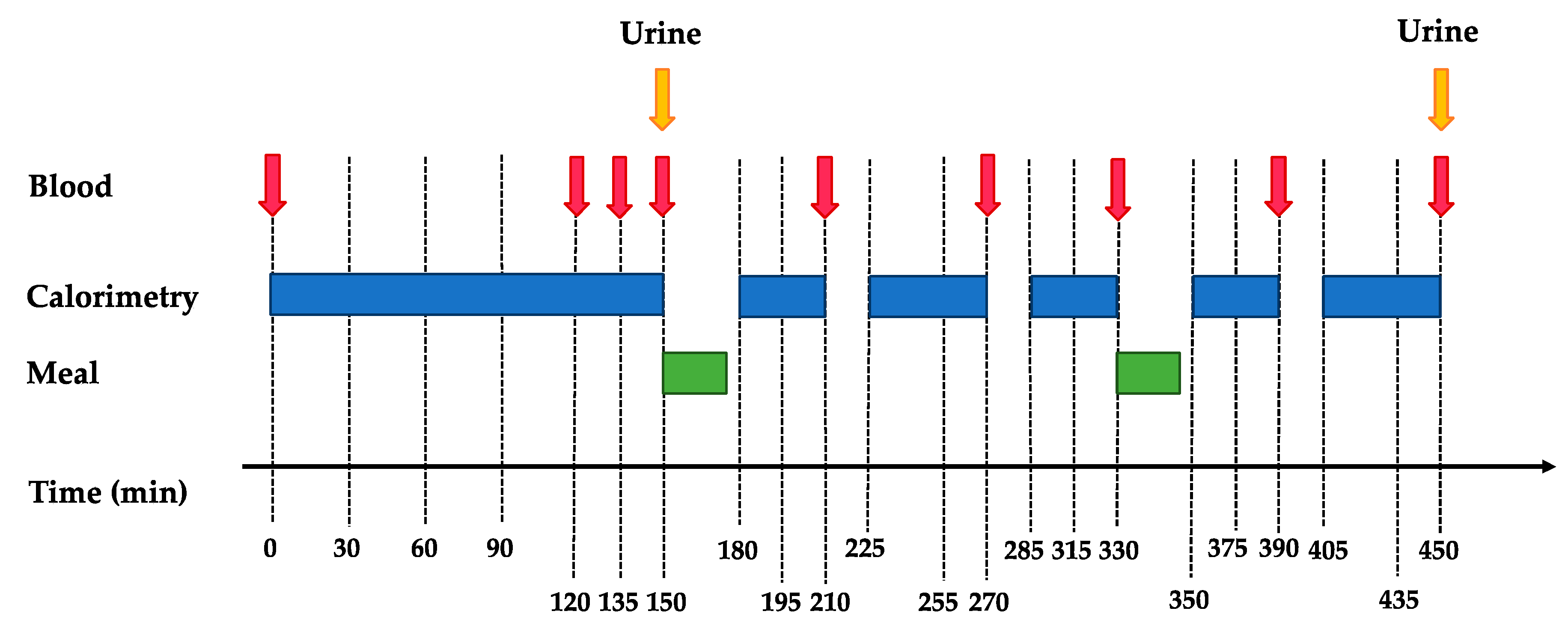
2.5. Metabolic Tests
2.6. Analytical Procedures
2.7. Statistical Analysis
3. Results
3.1. Fasting Condition
3.2. IHCL and IMCL Concentrations
3.3. Postprandial Parameters
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Szendroedi, J.; Roden, M. Ectopic lipids and organ function. Curr. Opin. Lipidol. 2009, 20, 50–56. [Google Scholar] [CrossRef]
- Morelli, M.; Gaggini, M.; Daniele, G.; Marraccini, P.; Sicari, R.; Gastaldelli, A. Ectopic fat: The true culprit linking obesity and cardiovascular disease? Thromb. Haemost. 2013, 110, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Britton, K.A.; Fox, C.S. Ectopic fat depots and cardiovascular disease. Circulation 2011, 124, e837–e841. [Google Scholar] [CrossRef] [PubMed]
- Stanhope, K.L.; Havel, P.J. Fructose consumption: Considerations for future research on its effects on adipose distribution, lipid metabolism, and insulin sensitivity in humans. J. Nutr. 2009, 139, 1236S–1241S. [Google Scholar] [CrossRef] [PubMed]
- Aeberli, I.; Hochuli, M.; Gerber, P.A.; Sze, L.; Murer, S.B.; Tappy, L.; Spinas, G.A.; Berneis, K. Moderate amounts of fructose consumption impair insulin sensitivity in healthy young men: A randomized controlled trial. Diabetes Care 2013, 36, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Stanhope, K.L.; Havel, P.J. Fructose consumption: Potential mechanisms for its effects to increase visceral adiposity and induce dyslipidemia and insulin resistance. Curr. Opin. Lipidol. 2008, 19, 16–24. [Google Scholar] [CrossRef]
- Stanhope, K.L.; Schwarz, J.M.; Keim, N.L.; Griffen, S.C.; Bremer, A.A.; Graham, J.L.; Hatcher, B.; Cox, C.L.; Dyachenko, A.; Zhang, W.; et al. Consuming fructose-sweetened, not glucose-sweetened, beverages increases visceral adiposity and lipids and decreases insulin sensitivity in overweight/obese humans. J. Clin. Investig. 2009, 119, 1322–1334. [Google Scholar] [CrossRef] [Green Version]
- Wang, D.D.; Sievenpiper, J.L.; de Souza, R.J.; Chiavaroli, L.; Ha, V.; Cozma, A.I.; Mirrahimi, A.; Yu, M.E.; Carleton, A.J.; Di Buono, M.; et al. The effects of fructose intake on serum uric acid vary among controlled dietary trials. J. Nutr. 2012, 142, 916–923. [Google Scholar] [CrossRef]
- Taskinen, M.R.; Soderlund, S.; Bogl, L.H.; Hakkarainen, A.; Matikainen, N.; Pietilainen, K.H.; Rasanen, S.; Lundbom, N.; Bjornson, E.; Eliasson, B.; et al. Adverse effects of fructose on cardiometabolic risk factors and hepatic lipid metabolism in subjects with abdominal obesity. J. Intern. Med. 2017, 282, 187–201. [Google Scholar] [CrossRef]
- Sobrecases, H.; Le, K.A.; Bortolotti, M.; Schneiter, P.; Ith, M.; Kreis, R.; Boesch, C.; Tappy, L. Effects of short-term overfeeding with fructose, fat and fructose plus fat on plasma and hepatic lipids in healthy men. Diabetes Metab. 2010, 36, 244–246. [Google Scholar] [CrossRef]
- Hamad, E.M.; Taha, S.H.; Abou Dawood, A.G.; Sitohy, M.Z.; Abdel-Hamid, M. Protective effect of whey proteins against nonalcoholic fatty liver in rats. Lipids Health Dis. 2011, 10, 57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chaumontet, C.; Even, P.C.; Schwarz, J.; Simonin-Foucault, A.; Piedcoq, J.; Fromentin, G.; Azzout-Marniche, D.; Tome, D. High dietary protein decreases fat deposition induced by high-fat and high-sucrose diet in rats. Br. J. Nutr. 2015, 114, 1132–1142. [Google Scholar] [CrossRef] [PubMed]
- Bortolotti, M.; Kreis, R.; Debard, C.; Cariou, B.; Faeh, D.; Chetiveaux, M.; Ith, M.; Vermathen, P.; Stefanoni, N.; Le, K.A.; et al. High protein intake reduces intrahepatocellular lipid deposition in humans. Am. J. Clin. Nutr. 2009, 90, 1002–1010. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martens, E.A.; Gatta-Cherifi, B.; Gonnissen, H.K.; Westerterp-Plantenga, M.S. The potential of a high protein-low carbohydrate diet to preserve intrahepatic triglyceride content in healthy humans. PLoS ONE 2014, 9, e109617. [Google Scholar] [CrossRef] [PubMed]
- Rietman, A.; Schwarz, J.; Blokker, B.A.; Siebelink, E.; Kok, F.J.; Afman, L.A.; Tome, D.; Mensink, M. Increasing protein intake modulates lipid metabolism in healthy young men and women consuming a high-fat hypercaloric diet. J. Nutr. 2014, 144, 1174–1180. [Google Scholar] [CrossRef] [PubMed]
- Theytaz, F.; Noguchi, Y.; Egli, L.; Campos, V.; Buehler, T.; Hodson, L.; Patterson, B.W.; Nishikata, N.; Kreis, R.; Mittendorfer, B.; et al. Effects of supplementation with essential amino acids on intrahepatic lipid concentrations during fructose overfeeding in humans. Am. J. Clin. Nutr. 2012, 96, 1008–1016. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bray, G.A.; Smith, S.R.; de Jonge, L.; Xie, H.; Rood, J.; Martin, C.K.; Most, M.; Brock, C.; Mancuso, S.; Redman, L.M. Effect of dietary protein content on weight gain, energy expenditure, and body composition during overeating: A randomized controlled trial. JAMA 2012, 307, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Eisenmann, A.; Amann, A.; Said, M.; Datta, B.; Ledochowski, M. Implementation and interpretation of hydrogen breath tests. J. Breath Res. 2008, 2, 046002. [Google Scholar] [CrossRef]
- Le, K.A.; Faeh, D.; Stettler, R.; Ith, M.; Kreis, R.; Vermathen, P.; Boesch, C.; Ravussin, E.; Tappy, L. A 4-wk high-fructose diet alters lipid metabolism without affecting insulin sensitivity or ectopic lipids in healthy humans. Am. J. Clin. Nutr. 2006, 84, 1374–1379. [Google Scholar] [CrossRef] [Green Version]
- Boesch, C.; Kreis, R. Observation of intramyocellular lipids by 1h-magnetic resonance spectroscopy. Ann. N. Y. Acad. Sci. 2000, 904, 25–31. [Google Scholar] [CrossRef]
- Cros, J.; Pianezzi, E.; Rosset, R.; Egli, L.; Schneiter, P.; Cornette, F.; Pouymayou, B.; Heinzer, R.; Tappy, L.; Kreis, R.; et al. Impact of sleep restriction on metabolic outcomes induced by overfeeding: A randomized controlled trial in healthy individuals. Am. J. Clin. Nutr. 2019. [Google Scholar] [CrossRef] [PubMed]
- Elia, M.; Livesey, G. Energy expenditure and fuel selection in biological systems: The theory and practice of calculations based on indirect calorimetry and tracer methods. World Rev. Nutr. Diet. 1992, 70, 68–131. [Google Scholar] [PubMed]
- Tran, C.; Jacot-Descombes, D.; Lecoultre, V.; Fielding, B.A.; Carrel, G.; Le, K.A.; Schneiter, P.; Bortolotti, M.; Frayn, K.N.; Tappy, L. Sex differences in lipid and glucose kinetics after ingestion of an acute oral fructose load. Br. J. Nutr. 2010, 104, 1139–1147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karpe, F.; Steiner, G.; Olivecrona, T.; Carlson, L.A.; Hamsten, A. Metabolism of triglyceride-rich lipoproteins during alimentary lipemia. J. Clin. Investig. 1993, 91, 748–758. [Google Scholar] [CrossRef] [PubMed]
- Lecoultre, V.; Egli, L.; Carrel, G.; Theytaz, F.; Kreis, R.; Schneiter, P.; Boss, A.; Zwygart, K.; Le, K.A.; Bortolotti, M.; et al. Effects of fructose and glucose overfeeding on hepatic insulin sensitivity and intrahepatic lipids in healthy humans. Obesity 2013, 21, 782–785. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnston, R.D.; Stephenson, M.C.; Crossland, H.; Cordon, S.M.; Palcidi, E.; Cox, E.F.; Taylor, M.A.; Aithal, G.P.; Macdonald, I.A. No difference between high-fructose and high-glucose diets on liver triacylglycerol or biochemistry in healthy overweight men. Gastroenterology 2013, 145, 1016–1025.e2. [Google Scholar] [CrossRef]
- Le, K.A.; Ith, M.; Kreis, R.; Faeh, D.; Bortolotti, M.; Tran, C.; Boesch, C.; Tappy, L. Fructose overconsumption causes dyslipidemia and ectopic lipid deposition in healthy subjects with and without a family history of type 2 diabetes. Am. J. Clin. Nutr. 2009, 89, 1760–1765. [Google Scholar] [CrossRef] [Green Version]
- Ngo Sock, E.T.; Le, K.A.; Ith, M.; Kreis, R.; Boesch, C.; Tappy, L. Effects of a short-term overfeeding with fructose or glucose in healthy young males. Br. J. Nutr. 2010, 103, 939–943. [Google Scholar] [CrossRef]
- Tappy, L.; Le, K.A. Does fructose consumption contribute to non-alcoholic fatty liver disease? Clin. Res. Hepatol. Gastroenterol. 2012, 36, 554–560. [Google Scholar] [CrossRef]
- Donnelly, K.L.; Smith, C.I.; Schwarzenberg, S.J.; Jessurun, J.; Boldt, M.D.; Parks, E.J. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. J. Clin. Investig. 2005, 115, 1343–1351. [Google Scholar] [CrossRef] [Green Version]
- Kotronen, A.; Yki-Jarvinen, H. Fatty liver: A novel component of the metabolic syndrome. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Ferramosca, A.; Zara, V. Modulation of hepatic steatosis by dietary fatty acids. World J. Gastroenterol. 2014, 20, 1746–1755. [Google Scholar] [CrossRef]
- Teff, K.L.; Elliott, S.S.; Tschop, M.; Kieffer, T.J.; Rader, D.; Heiman, M.; Townsend, R.R.; Keim, N.L.; D’Alessio, D.; Havel, P.J. Dietary fructose reduces circulating insulin and leptin, attenuates postprandial suppression of ghrelin, and increases triglycerides in women. J. Clin. Endocrinol. Metab. 2004, 89, 2963–2972. [Google Scholar] [CrossRef] [PubMed]
- Stanhope, K.L.; Bremer, A.A.; Medici, V.; Nakajima, K.; Ito, Y.; Nakano, T.; Chen, G.; Fong, T.H.; Lee, V.; Menorca, R.I.; et al. Consumption of fructose and high fructose corn syrup increase postprandial triglycerides, ldl-cholesterol, and apolipoprotein-b in young men and women. J. Clin. Endocrinol. Metab. 2011, 96, E1596–E1605. [Google Scholar] [CrossRef] [PubMed]
- Tappy, L. Thermic effect of food and sympathetic nervous system activity in humans. Reprod. Nutr. Dev. 1996, 36, 391–397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calbet, J.A.; MacLean, D.A. Plasma glucagon and insulin responses depend on the rate of appearance of amino acids after ingestion of different protein solutions in humans. J. Nutr. 2002, 132, 2174–2182. [Google Scholar] [CrossRef] [PubMed]
- Ramnanan, C.J.; Edgerton, D.S.; Kraft, G.; Cherrington, A.D. Physiologic action of glucagon on liver glucose metabolism. Diabetes Obes. Metab. 2011, 13 (Suppl. 1), 118–125. [Google Scholar] [CrossRef] [Green Version]
- Granger, D.N.; Richardson, P.D.; Kvietys, P.R.; Mortillaro, N.A. Intestinal blood flow. Gastroenterology 1980, 78, 837–863. [Google Scholar] [PubMed]
- Ma, J.; Stevens, J.E.; Cukier, K.; Maddox, A.F.; Wishart, J.M.; Jones, K.L.; Clifton, P.M.; Horowitz, M.; Rayner, C.K. Effects of a protein preload on gastric emptying, glycemia, and gut hormones after a carbohydrate meal in diet-controlled type 2 diabetes. Diabetes Care 2009, 32, 1600–1602. [Google Scholar] [CrossRef] [PubMed]
- Le, M.T.; Frye, R.F.; Rivard, C.J.; Cheng, J.; McFann, K.K.; Segal, M.S.; Johnson, R.J.; Johnson, J.A. Effects of high-fructose corn syrup and sucrose on the pharmacokinetics of fructose and acute metabolic and hemodynamic responses in healthy subjects. Metabolism 2012, 61, 641–651. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahloulay, M.; Dechaux, M.; Laborde, K.; Bankir, L. Influence of glucagon on gfr and on urea and electrolyte excretion: Direct and indirect effects. Am. J. Physiol. 1995, 269, F225–F235. [Google Scholar] [CrossRef] [PubMed]
- Yu, T.F.; Sirota, J.H.; Berger, L.; Halpern, M.; Gutman, A.B. Effect of sodium lactate infusion on urate clearance in man. Proc. Soc. Exp. Biol. Med. 1957, 96, 809–813. [Google Scholar] [CrossRef] [PubMed]
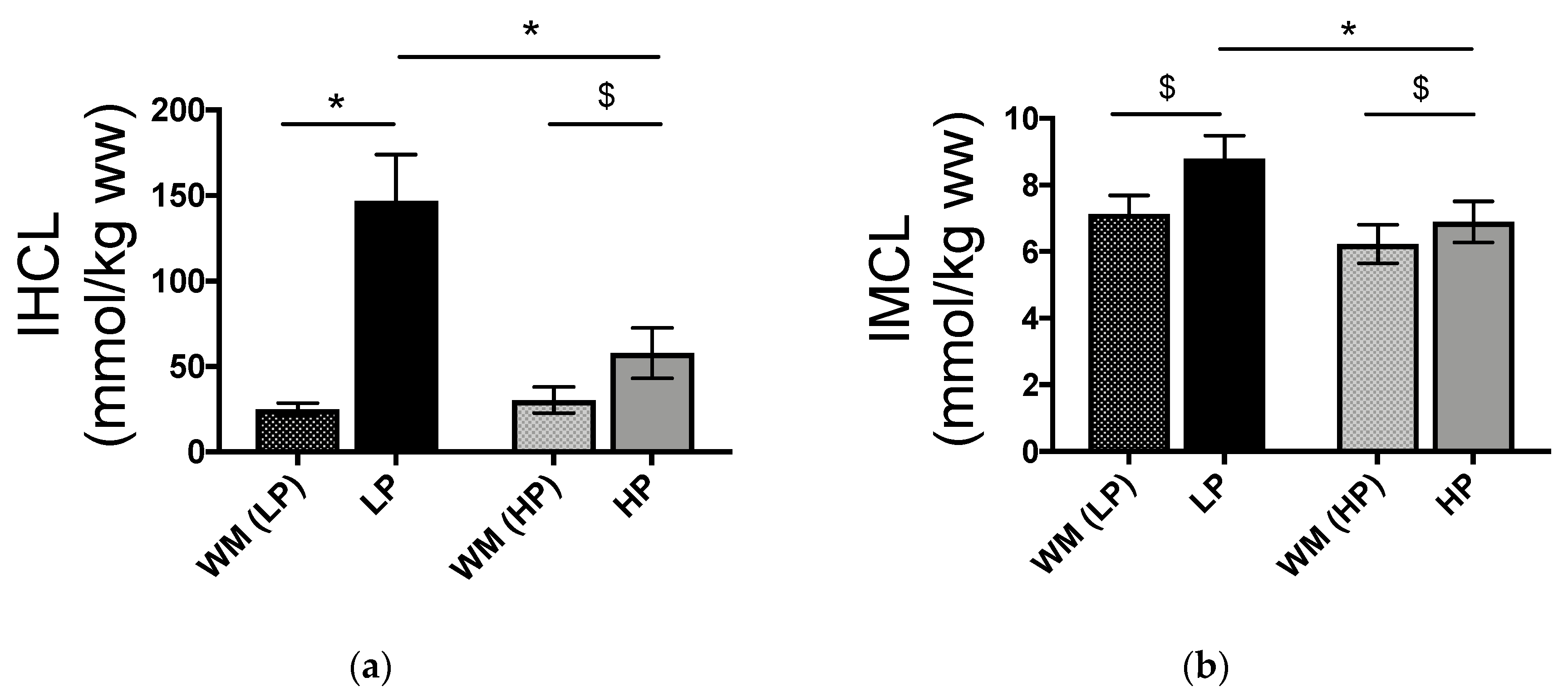
| Diet Composition | WM | LP-HF | HP-LF | |||||
|---|---|---|---|---|---|---|---|---|
| Solid Diet kcal/day (%) | Beverages kcal/day (%) | Solid Diet kcal/day (%) | Beverages kcal/day (%) | Total LP-HF kcal/day (%) | Solid Diet kcal/day (%) | Beverages kcal/day (%) | Total HP-LF kcal/day (%) | |
| Starch | 1061 (45) | - | 1054 | - | 1054 (29) | 1043 | - | 1043 (29) |
| Sucrose | 249 (10) | - | 241 | 965 | 1206 (34) | 246 | 964 | 1210 (34) |
| Lactose | - | - | - | 245 | 245 (7) | - | 246 | 246 (7) |
| Protein | 274 (12) | - | 194 | - | 194 (5) | 514 | 178 | 692 (20) |
| Fat | 781 (33) | - | 886 | - | 886 (25) | 357 | 12 | 369 (10) |
| SFA | 263 (34) | - | 313 | - | 313 (35) | 184 | - | 184 (52) |
| MUFA | 280 (36) | - | 389 | - | 389 (44) | 102 | - | 102 (29) |
| PUFA | 202 (26) | - | 168 | - | 168 (19) | 55 | - | 55 (15) |
| Total kcal | 2365 | - | 2375 | 1210 | 3585 | 2160 | 1400 | 3560 |
| Fasting | WM (LP-HF) | LP-HF | WM (HP-LF) | HP-LF | p Value | ||
|---|---|---|---|---|---|---|---|
| Overfeeding | Protein/Fat Content | OxP | |||||
| Glucose (mmol/L) | 4.56 ± 0.07 | 4.78 ± 0.07 | 4.46 ± 0.11 | 4.76 ± 0.09 | <0.001 | 0.444 | 0.383 |
| Fructose (μmol/L) | 25.95 ± 1.41 | 27.15 ± 1.49 | 26.35 ± 1.37 | 28.0 ± 1.32 | 0.022 | 0.611 | 0.750 |
| Lactate (mmol/L) | 0.70 ± 0.06 | 1.22 ± 0.07 | 0.64 ± 0.04 | 1.16 ± 0.09 | <0.001 | 0.107 | 0.935 |
| Uric acid (mmol/L) | 0.38 ± 0.02 | 0.38 ± 0.03 | 0.39 ± 0.02 | 0.30 ± 0.02 | <0.001 | <0.001 | <0.001 |
| TG (mmol/L) | 0.68 ± 0.07 | 1.54 ± 0.22 | 0.66 ± 0.08 | 1.68 ± 0.19 | <0.001 | 0.429 | 0.119 |
| NEFA (mmol/L) | 0.72 ± 0.05 | 0.44 ± 0.09 | 0.77 ± 0.04 | 0.37 ± 0.07 | <0.001 | 0.779 | 0.097 |
| Insulin (μU/mL) | 8.42 ± 0.83 | 10.95 ± 1.02 | 7.82 ± 0.79 | 11.73 ± 1.54 | <0.001 | 0.744 | 0.295 |
| Glucagon (pg/mL) | 72.42 ± 4.83 | 72.49 ± 4.94 | 68.67 ± 4.03 | 79.27 ± 5.35 | 0.059 | 0.276 | 0.036 |
| IGF-1 (ng/mL) | 212 ± 13 | 176 ± 12 | 174 ± 18 | 208 ± 13 | 0.901 | 0.712 | <0.001 |
| Postprandial | WM (LP-HF) | LP-HF | WM (HP-LF) | HP-LF | p Value | ||
|---|---|---|---|---|---|---|---|
| Overfeeding | Protein/Fat Content | OxP | |||||
| iAUC Glucose (mmol/L*300min) | 504.0 ± 40.5 | 495.3 ± 69.2 | 560.3 ± 43.7 | 471.4 ± 57.2 | 0.242 | 0.616 | 0.189 |
| iAUC Fructose (mmol/L*300min) | 4.2 ± 0.3 | 30.3 ± 2.9 | 4.8 ± 0.5 | 23.4 ± 2.2 | <0.001 | 0.005 | 0.003 |
| iAUC Lactate (mmol/L*300min) | 78.8 ± 12.8 | 239.8 ± 24.5 | 92.2 ± 15.1 | 139.3 ± 15.7 | <0.001 | 0.001 | 0.001 |
| iAUC TG (mmol/L*300min) | 29.3 ± 6.8 | 121.3 ± 15.3 | 24.2 ± 8.0 | 126.7 ± 16.9 | <0.001 | 0.986 | 0.471 |
| iAUC NEFA (mmol/L*300min) | −162.6 ± 12.5 | −86.7 ± 23.2 | −173.1 ± 11.0 | −76.6 ± 17.2 | <0.001 | 0.984 | 0.051 |
| iAUC Insulin (μU/mL*300min) | 11378 ± 1232 | 19228 ± 1708 | 11138 ± 1488 | 24123 ± 2790 | <0.001 | 0.061 | 0.028 |
| WM (LP-HF) | WM (HP-LF) | LP-HF | HP-LF | p Value | |||
|---|---|---|---|---|---|---|---|
| Overfeeding | Protein/Fat Content | OxP | |||||
| 24-h urinary excretion | |||||||
| Creatinine (mmol/24h) | 13.6 ± 1.8 | 12.6 ± 1.2 | 13.0 ± 0.8 | 12.7 ± 1.1 | 0.264 | 0.450 | 0.638 |
| Uric acid (mmol/24h) | 3.5 ± 0.2 | 3.3 ± 0.2 | 3.3 ± 0.2 | 4.1 ± 0.4 | 0.049 | 0.238 | 0.022 |
| Urinary clearance rate | |||||||
| Creatinine (ml/min) | 129.8 ± 9.4 | 133.7 ± 9.7 | 135.1 ± 10.8 | 153.0 ± 13.0 | 0.279 | 0.309 | 0.282 |
| Uric acid (ml/min) | 6.9 ± 0.6 | 6.5 ± 0.6 | 6.1 ± 0.4 | 10.0 ± 1.4 | 0.005 | 0.015 | 0.004 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Surowska, A.; Jegatheesan, P.; Campos, V.; Marques, A.-S.; Egli, L.; Cros, J.; Rosset, R.; Lecoultre, V.; Kreis, R.; Boesch, C.; et al. Effects of Dietary Protein and Fat Content on Intrahepatocellular and Intramyocellular Lipids during a 6-Day Hypercaloric, High Sucrose Diet: A Randomized Controlled Trial in Normal Weight Healthy Subjects. Nutrients 2019, 11, 209. https://0-doi-org.brum.beds.ac.uk/10.3390/nu11010209
Surowska A, Jegatheesan P, Campos V, Marques A-S, Egli L, Cros J, Rosset R, Lecoultre V, Kreis R, Boesch C, et al. Effects of Dietary Protein and Fat Content on Intrahepatocellular and Intramyocellular Lipids during a 6-Day Hypercaloric, High Sucrose Diet: A Randomized Controlled Trial in Normal Weight Healthy Subjects. Nutrients. 2019; 11(1):209. https://0-doi-org.brum.beds.ac.uk/10.3390/nu11010209
Chicago/Turabian StyleSurowska, Anna, Prasanthi Jegatheesan, Vanessa Campos, Anne-Sophie Marques, Léonie Egli, Jérémy Cros, Robin Rosset, Virgile Lecoultre, Roland Kreis, Chris Boesch, and et al. 2019. "Effects of Dietary Protein and Fat Content on Intrahepatocellular and Intramyocellular Lipids during a 6-Day Hypercaloric, High Sucrose Diet: A Randomized Controlled Trial in Normal Weight Healthy Subjects" Nutrients 11, no. 1: 209. https://0-doi-org.brum.beds.ac.uk/10.3390/nu11010209





