Staphylococcus aureus Exotoxins and Their Detection in the Dairy Industry and Mastitis
Abstract
:1. Introduction
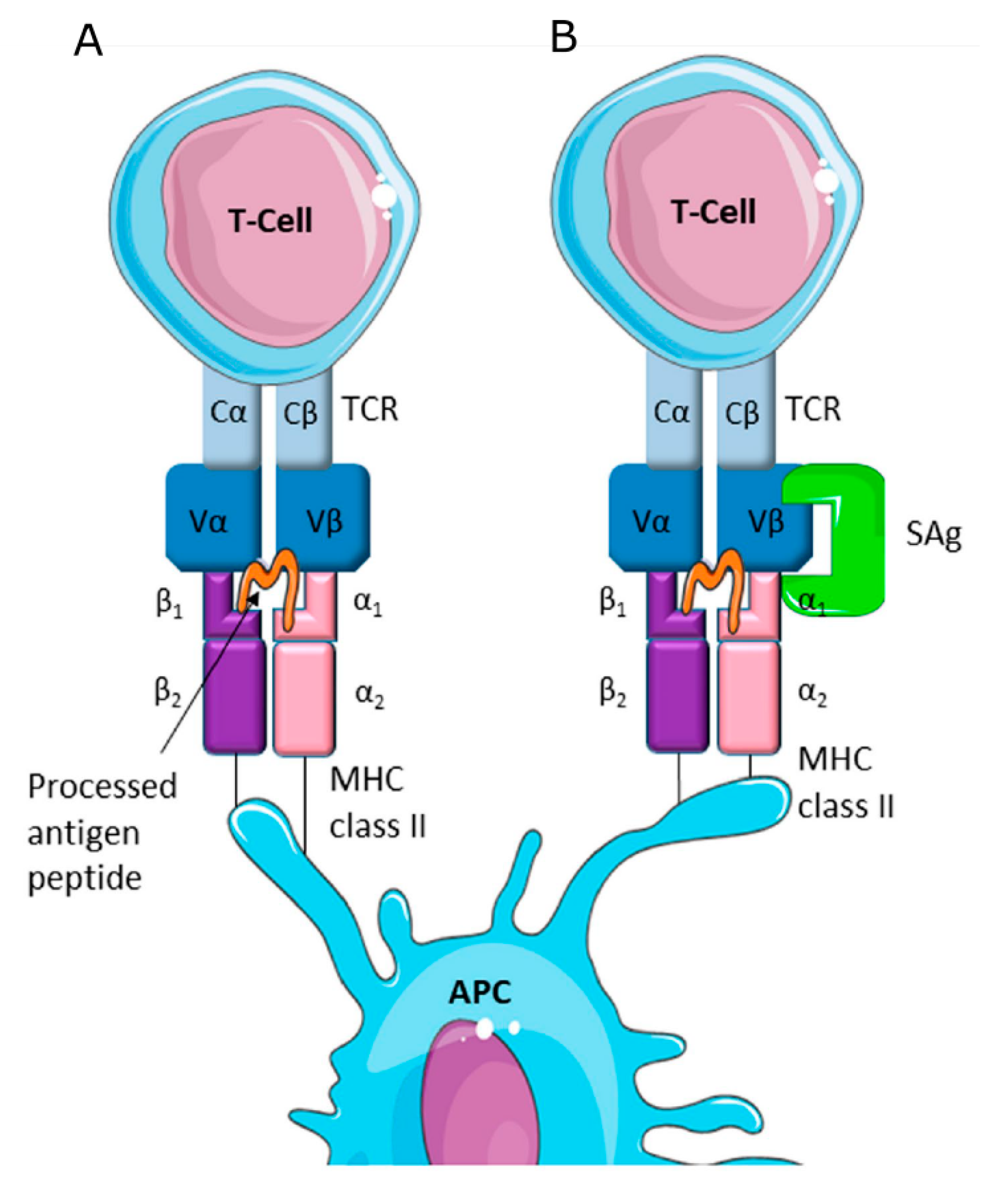
2. Role of Staphylococcus aureus Toxic Proteins in Mastitis
3. Staphylococcus aureus Exotoxins in Food
4. Staphylococcus aureus Exotoxin Identification in Food and Milk
4.1. Staphylococcus aureus Toxin Identification Using Conventional Methods
4.2. Staphylococcus aureus Toxin Identification Using Genomic Methods
4.3. Staphylococcus aureus Toxin Identification Using Immunoassays
4.4. New Trends in Staphylococcus aureus Toxin Identification: Aptamers, Molecularly Imprinted Polymers, Proteomics, and Next-Generation Sequencing
5. Toxins in S. aureus Treatment
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Merz, A.; Stephan, R.; Johler, S. Staphylococcus aureus Isolates from goat and sheep milk seem to be closely related and differ from isolates detected from bovine milk. Front. Microbiol. 2016, 7, 319. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, D.; Zhang, L.; Yong, C.; Shen, M.; Ali, T.; Shahid, M.; Han, K.; Zhou, X.; Han, B. Relationships among superantigen toxin gene profiles, genotypes, and pathogenic characteristics of Staphylococcus aureus isolates from bovine mastitis. J. Dairy Sci. 2017, 100, 4276–4286. [Google Scholar] [CrossRef] [PubMed]
- Tuffs, S.W.; Haeryfar, S.M.M.; McCormick, J.K. Manipulation of Innate and Adaptive Immunity by Staphylococcal Superantigens. Pathogens 2018, 7, 53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, W.; Lin, X.; Jiang, T.; Peng, Z.; Xu, J.; Yi, L.; Li, F.; Fanning, S.; Baloch, Z. Prevalence and characterization of Staphylococcus aureus cultured from raw milk taken from dairy cows with mastitis in Beijing, China. Front. Microbiol. 2018, 9, 1123. [Google Scholar] [CrossRef] [PubMed]
- Schwan, W.R. Staphylococcus aureus Toxins: Armaments for a Significant Pathogen. Toxins 2019, 11, 457. [Google Scholar] [CrossRef] [Green Version]
- Imanishi, I.; Nicolas, A.; Caetano, A.C.B.; de Castro, T.L.P.; Tartaglia, N.R.; Mariutti, R.; Guédon, E.; Even, S.; Berkova, N.; Arni, R.K.; et al. Exfoliative toxin E, a new Staphylococcus aureus virulence factor with host-specific activity. Sci. Rep. 2019, 9, 16336. [Google Scholar] [CrossRef]
- Ageitos, J.M.; Sánchez-Pérez, A.; Calo-Mata, P.; Villa, T.G. Antimicrobial peptides (AMPs): Ancient compounds that represent novel weapons in the fight against bacteria. Biochem. Pharmacol. 2017, 133, 117–138. [Google Scholar] [CrossRef]
- Reddy, P.N.; Srirama, K.; Dirisala, V.R. An Update on Clinical burden, diagnostic tools, and therapeutic options of Staphylococcus aureus. Infect. Dis. Res. Treat. 2017, 10, 117991611770399. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, D.; Borges, A.; Simões, M. Staphylococcus aureus toxins and their molecular activity in infectious diseases. Toxins 2018, 10, 252. [Google Scholar] [CrossRef] [Green Version]
- Wang, F.; Hongjun, Y.; Hong-Bin, H.; Changfa, W.; Yundong, G.; Qifeng, Z.; Xiaohong, W.; Yanjun, Z. Study on the hemolysin phenotype and the genetype distribution of staphyloccocus aureus caused bovine mastitis in shandong dairy farms. Int. J. Appl. Res. Vet. Med. 2011, 9, 416–421. [Google Scholar]
- Denayer, S.; Delbrassinne, L.; Nia, Y.; Botteldoorn, N. Food-borne outbreak investigation and molecular typing: High diversity of Staphylococcus aureus strains and importance of toxin detection. Toxins 2017, 9, 407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fueyo, J.M.; Mendoza, M.C.; Rodicio, M.R.; Muñiz, J.; Alvarez, M.A.; Martín, M.C. Cytotoxin and pyrogenic toxin superantigen gene profiles of Staphylococcus aureus associated with subclinical mastitis in dairy cows and relationships with macrorestriction genomic profiles. J. Clin. Microbiol. 2005, 43, 1278–1284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vaughn, J.M.; Abdi, R.D.; Gillespie, B.E.; Kerro Dego, O. Genetic diversity and virulence characteristics of Staphylococcus aureus isolates from cases of bovine mastitis. Microb. Pathog. 2020, 144, 104171. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Huseby, D.L.; Brandis, G.; Hughes, D. Alternative evolutionary pathways for drug-resistant small colony variant mutants in Staphylococcus aureus. MBio 2017, 8, 3. [Google Scholar] [CrossRef] [Green Version]
- Akineden, Ö.; Annemüller, C.; Hassan, A.A.; Lämmler, C.; Wolter, W.; Zschöck, M. Toxin genes and other characteristics of Staphylococcus aureus isolates from milk of cows with mastitis. Clin. Diagn. Lab. Immunol. 2001, 8, 959–964. [Google Scholar] [CrossRef] [Green Version]
- Stephan, R.; Annemüller, C.; Hassan, A.A.; Lämmler, C. Characterization of enterotoxigenic Staphylococcus aureus strains isolated from bovine mastitis in north-east Switzerland. Vet. Microbiol. 2001, 78, 373–382. [Google Scholar] [CrossRef]
- Hoekstra, J.; Rutten, V.; Sommeling, L.; van Werven, T.; Spaninks, M.; Duim, B.; Benedictus, L.; Koop, G. High Production of LukMF’ in Staphylococcus aureus Field strains is associated with clinical bovine mastitis. Toxins 2018, 10, 200. [Google Scholar] [CrossRef] [Green Version]
- Srinivasan, V.; Sawant, A.A.; Gillespie, B.E.; Headrick, S.J.; Ceasaris, L.; Oliver, S.P. Prevalence of enterotoxin and toxic shock syndrome toxin genes in Staphylococcus aureus Isolated from Milk of Cows with Mastitis. Foodborne Pathog. Dis. 2006, 3, 274–283. [Google Scholar] [CrossRef]
- Rainard, P.; Corrales, J.C.; Barrio, M.B.; Cochard, T.; Poutrel, B. Leucotoxic activities of Staphylococcus aureus strains isolated from cows, ewes, and goats with mastitis: Importance of LukM/LukF′-PV leukotoxin. Clin. Diagn. Lab. Immunol. 2003, 10, 272–277. [Google Scholar] [CrossRef] [Green Version]
- Costa, F.N.; Belo, N.O.; Costa, E.A.; Andrade, G.I.; Pereira, L.S.; Carvalho, I.A.; Santos, R.L. Frequency of enterotoxins, toxic shock syndrome toxin-1, and biofilm formation genes in Staphylococcus aureus isolates from cows with mastitis in the Northeast of Brazil. Trop. Anim. Health Prod. 2018, 50, 1089–1097. [Google Scholar] [CrossRef]
- Vimercati, C.; Cremonesi, P.; Castiglioni, B.; Pisoni, G.; Boettcher, P.J.; Stella, A.; Vicenzoni, G.; Moroni, P. Molecular Typing of Staphylococcus aureus Isolated from cows, goats and sheep with intramammary infections on the basis of gene polymorphisms and toxins genes. J. Vet. Med. Ser. B 2006, 53, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Orden, J.A.; Goyache, J.; Hernández, J.; Doménech, A.; Suárez, G.; Gómez-Lucia, E. Detection of enterotoxins and TSST-1 secreted by Staphylococcus aureus isolated from ruminant mastitis. Comparison of ELISA and immunoblot. J. Appl. Bacteriol. 1992, 72, 486–489. [Google Scholar] [CrossRef] [PubMed]
- Mørk, T.; Tollersrud, T.; Kvitle, B.; Jørgensen, H.J.; Waage, S. Comparison of Staphylococcus aureus genotypes recovered from cases of bovine, ovine, and caprine mastitis. J. Clin. Microbiol. 2005, 43, 3979–3984. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, L.; Gao, J.; Barkema, H.W.; Ali, T.; Liu, G.; Deng, Y.; Naushad, S.; Kastelic, J.P.; Han, B. Virulence gene profiles: Alpha-hemolysin and clonal diversity in Staphylococcus aureus isolates from bovine clinical mastitis in China. BMC Vet. Res. 2018, 14, 63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mello, P.; Moraes Riboli, D.; Pinheiro, L.; de Almeida Martins, L.; Vasconcelos Paiva Brito, M.; Ribeiro de Souza da Cunha, M. Detection of enterotoxigenic potential and determination of clonal profile in Staphylococcus aureus and coagulase-negative staphylococci isolated from bovine subclinical mastitis in different brazilian states. Toxins 2016, 8, 104. [Google Scholar] [CrossRef] [Green Version]
- Fursova, K.K.; Shchannikova, M.P.; Loskutova, I.V.; Shepelyakovskaya, A.O.; Laman, A.G.; Boutanaev, A.M.; Sokolov, S.L.; Artem’eva, O.A.; Nikanova, D.A.; Zinovieva, N.A.; et al. Exotoxin diversity of Staphylococcus aureus isolated from milk of cows with subclinical mastitis in Central Russia. J. Dairy Sci. 2018, 101, 4325–4331. [Google Scholar] [CrossRef] [Green Version]
- Mclauchlin, J.; Narayanan, G.L.; Mithani, V.; O’Neill, G. The detection of enterotoxins and toxic shock syndrome toxin genes in Staphylococcus aureus by polymerase chain reaction. J. Food Prot. 2000, 63, 479–488. [Google Scholar] [CrossRef]
- Mansour, A.S.; Wagih, G.E.S.; Morgan, S.D.; Elhariri, M.; El-Shabrawy, M.A.; Abuelnaga, A.S.M.; Elgabry, E.A. Detection of Staphylococcus aureus enterotoxigenic strains in bovine raw milk by reversed passive latex agglutination and multiplex polymerase chain reaction. Vet. World 2017, 10, 843–847. [Google Scholar] [CrossRef]
- Fujikawa, H.; Igarashi, H. Rapid latex agglutination test for detection of staphylococcal enterotoxins A to E that uses high-density latex particles. Appl. Environ. Microbiol. 1988, 54, 2345–2348. [Google Scholar] [CrossRef] [Green Version]
- Kong, C.; Neoh, H.M.; Nathan, S. Targeting Staphylococcus aureus toxins: A potential form of anti-virulence therapy. Toxins 2016, 8, 72. [Google Scholar] [CrossRef] [Green Version]
- Seilie, E.S.; Bubeck Wardenburg, J. Staphylococcus aureus pore-forming toxins: The interface of pathogen and host complexity. Semin. Cell Dev. Biol. 2017, 72, 101–116. [Google Scholar] [CrossRef]
- Barrio, M.B.; Rainard, P.; Prévost, G. LukM/LukF′-PV is the most active Staphylococcus aureus leukotoxin on bovine neutrophils. Microbes Infect. 2006, 8, 2068–2074. [Google Scholar] [CrossRef] [PubMed]
- Adkins, P.R.F.; Middleton, J.R.; Fox, L.K. Comparison of Virulence Gene Identification, Ribosomal Spacer PCR, and Pulsed-Field Gel Electrophoresis for Typing of Staphylococcus aureus strains isolated from cases of subclinical bovine mastitis in the united states. J. Clin. Microbiol. 2016, 54, 1871–1876. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, S.; Duan, N.; Gu, H.; Hao, L.; Ye, H.; Gong, W.; Wang, Z. A Review of the methods for detection of Staphylococcus aureus Enterotoxins. Toxins 2016, 8, 176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schubert, J.; Podkowik, M.; Bystroń, J.; Bania, J. Production of staphylococcal enterotoxins D and R in milk and meat juice by Staphylococcus aureus strains. Foodborne Pathog. Dis. 2017, 14, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.T.M.; Oliveira, D.S.V.; Veloso, V.S.; Silva, S.D.S.P.; Santos, L.S.; Neto, A.F.L.; Oliveira, F.A.D.A.; De Melo, M.C.N.; Soares, M.J.D.S. Microbiology quality, detection of enterotoxin genes and antimicrobial resistance of Staphylococcus aureus isolated from milk and Coalho cheese. Semin. Agrar. 2018, 39, 1957–1968. [Google Scholar] [CrossRef]
- Babic, M.; Pajić, M.; Nikolić, A.; Teodorović, V.; Mirilović, M.; Milojević, L.; Velebit, B. Expression of toxic shock syndrome toxin-1 gene of Staphylococcus aureus in milk: Proof of concept. Staphylococcus aureus in milk. Mljekarstvo 2018, 68, 12–20. [Google Scholar] [CrossRef]
- Ono, H.K.; Sato’o, Y.; Narita, K.; Naito, I.; Hirose, S.; Hisatsune, J.; Asano, K.; Hu, D.L.; Omoe, K.; Sugai, M.; et al. Identification and characterization of a novel staphylococcal emetic toxin. Appl. Environ. Microbiol. 2015, 81, 7034–7040. [Google Scholar] [CrossRef] [Green Version]
- Bhatia, A.; Zahoor, S. Staphylococcus aureus Enterotoxins: A Review. 2007, Volume 1. Available online: http://www.jcdr.net/back_issues.asp?issn=0973-709x&year=2007&month=June&volume=1&issue=3&page=188-197&id=52 (accessed on 4 June 2007).
- Kahya, S.; Guran, H.S.; Yilmaz, O. PCR and ELISA for staphylococcal enterotoxins and detection of some exotoxins from Staphylococcus spp. strains by PCR. Med. Weter. 2016, 72, 28–33. [Google Scholar]
- Badiou, C.; Dumitrescu, O.; George, N.; Forbes, A.R.N.; Drougka, E.; Chan, K.S.; Ramdani-Bouguessa, N.; Meugnier, H.; Bes, M.; Vandenesch, F.; et al. Rapid detection of Staphylococcus aureus Panton-Valentine Leukocidin in clinical specimens by enzyme-linked immunosorbent assay and immunochromatographic tests. J. Clin. Microbiol. 2010, 48, 1384–1390. [Google Scholar] [CrossRef] [Green Version]
- Lina, G.; Piemont, Y.; Godail-Gamot, F.; Bes, M.; Peter, M.-O.; Gauduchon, V.; Vandenesch, F.; Etienne, J. Involvement of Panton-Valentine Leukocidin—Producing Staphylococcus aureus in primary skin infections and pneumonia. Clin. Infect. Dis. 1999, 29, 1128–1132. [Google Scholar] [CrossRef] [PubMed]
- Johnsson, D.; Mölling, P.; Strålin, K.; Söderquist, B. Detection of Panton–Valentine leukocidin gene in Staphylococcus aureus by LightCycler PCR: Clinical and epidemiological aspects. Clin. Microbiol. Infect. 2004, 10, 884–889. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McClure, J.A.; Conly, J.M.; Lau, V.; Elsayed, S.; Louie, T.; Hutchins, W.; Zhang, K. Novel multiplex PCR assay for detection of the staphylococcal virulence marker Panton-Valentine leukocidin genes and simultaneous discrimination of methicillin-susceptible from-resistant staphylococci. J. Clin. Microbiol. 2006, 44, 1141–1144. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vane, S.C.S.; Prakasam, G. Detection of etaB gene for the production of exfoliative toxin among clinical isolates of Staphylococcus aureus. Res. J. Pharm. Technol. 2016, 9, 1469–1471. [Google Scholar] [CrossRef]
- Lacoma De La Torre, A.; Laabei, M.; Plans, O.; Martinez Vega, S.; Gomes-Fernandes, M.; Arméstar, F.; Villanueva, C.; Ricart, P.; Prat, C. Detection of alpha-hemolysin in Staphylococcus aureus derived respiratory strains: Correlation with microbiological and clinical variables. Eur. Resp. J. 2017, 50, PA4081. [Google Scholar] [CrossRef]
- Sospedra, I.; Soler, C.; Mañes, J.; Soriano, J.M. Rapid whole protein quantitation of staphylococcal enterotoxins A and B by liquid chromatography/mass spectrometry. J. Chromatogr. A 2012, 1238, 54–59. [Google Scholar] [CrossRef]
- Koike, H.; Kanda, M.; Hayashi, H.; Matsushima, Y.; Ohba, Y.; Nakagawa, Y.; Nagano, C.; Sekimura, K.; Hirai, A.; Shindo, T.; et al. Quantification of staphylococcal enterotoxin type A in cow milk by using a stable isotope-labelled peptide via liquid chromatography–tandem mass spectrometry. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2019, 36, 1098–1108. [Google Scholar] [CrossRef]
- Wieneke, A.A. The detection of enterotoxin and toxic shock syndrome toxin-1 production by strains of Staphylococcus aureus with commercial RPLA kits. Int. J. Food Microbiol. 1988, 7, 25–30. [Google Scholar] [CrossRef]
- Oishi, K.; Baba, T.; Nakatomi, Y.; Ito, T.; Hiramatsu, K. A latex agglutination assay for specific detection of Panton-Valentine leukocidin. J. Microbiol. Methods 2008, 75, 411–415. [Google Scholar] [CrossRef]
- Løvseth, A.; Loncarevic, S.; Berdal, K.G. Modified multiplex PCR method for detection of pyrogenic exotoxin genes in staphylococcal isolates. J. Clin. Microbiol. 2004, 42, 3869–3872. [Google Scholar] [CrossRef] [Green Version]
- Mahfoozi, A.; Shirzad-Aski, H.; Kaboosi, H.; Ghaemi, E.A. Identification of the classical enterotoxin genes of Staphylococcus aureus in various foods by multiplex PCR assay. Iran. J. Vet. Res. 2019, 20, 209. [Google Scholar] [PubMed]
- Becker, K.; Roth, R.; Peters, G. Rapid and specific detection of toxigenic Staphylococcus aureus: Use of two multiplex PCR enzyme immunoassays for amplification and hybridization of staphylococcal enterotoxin genes, exfoliative toxin genes, and toxic shock syndrome toxin 1 gene. J. Clin. Microbiol. 1998, 36, 2548–2553. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Law, J.W.F.; Mutalib, N.S.A.; Chan, K.G.; Lee, L.H. Rapid methods for the detection of foodborne bacterial pathogens: Principles, applications, advantages and limitations. Front. Microbiol. 2014, 5, 770. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cremonesi, P.; Luzzana, M.; Brasca, M.; Morandi, S.; Lodi, R.; Vimercati, C.; Agnellini, D.; Caramenti, G.; Moroni, P.; Castiglioni, B. Development of a multiplex PCR assay for the identification of Staphylococcus aureus enterotoxigenic strains isolated from milk and dairy products. Mol. Cell. Probes 2005, 19, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Xia, D.; Ma, P.; Gao, X.; Kang, W.; Wei, J. Advances in the detection of virulence genes of Staphylococcus aureus originate from food. Food Sci. Hum. Wellness 2020, 9, 40–44. [Google Scholar] [CrossRef]
- Schuberth, H.J.; Krueger, C.; Zerbe, H.; Bleckmann, E.; Leibold, W. Characterization of leukocytotoxic and superantigen-like factors produced by Staphylococcus aureus isolates from milk of cows with mastitis. Vet. Microbiol. 2001, 82, 187–199. [Google Scholar] [CrossRef]
- Umesha, S.; Manukumar, H.M. Advanced molecular diagnostic techniques for detection of food-borne pathogens: Current applications and future challenges. Crit. Rev. Food Sci. Nutr. 2018, 58, 84–104. [Google Scholar] [CrossRef]
- Kasai, S.; Yokota, A.; Zhou, H.; Nishizawa, M.; Niwa, K.; Onouchi, T.; Matsue, T. Immunoassay of the MRSA-related toxic protein, leukocidin, with scanning electrochemical microscopy. Anal. Chem. 2000, 72, 5761–5765. [Google Scholar] [CrossRef]
- Loeffler, D.A.; Creasy, M.T.; Norcross, N.L.; Paape, M.J. Enzyme-linked immunosorbent assay for detection of leukocidin toxin from Staphylococcus aureus in bovine milk samples. J. Clin. Microbiol. 1988, 26, 1331–1334. [Google Scholar] [CrossRef] [Green Version]
- Shahdordizadeh, M.; Taghdisi, S.M.; Ansari, N.; Alebooye Langroodi, F.; Abnous, K.; Ramezani, M. Aptamer based biosensors for detection of Staphylococcus aureus. Sens. Actuators B Chem. 2017, 241, 619–635. [Google Scholar] [CrossRef]
- Ahari, H.; Hedayati, M.; Akbari-adergani, B.; Kakoolaki, S.; Hosseini, H.; Anvar, A. Staphylococcus aureus exotoxin detection using potentiometric nanobiosensor for microbial electrode approach with the effects of pH and temperature. Int. J. Food Prop. 2017, 20, 1578–1587. [Google Scholar] [CrossRef]
- Cheng, K.; Chui, H.; Domish, L.; Hernandez, D.; Wang, G. Recent development of mass spectrometry and proteomics applications in identification and typing. PROTEOMICS–Clin. Appl. 2016, 10, 346–357. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.J. Food Forensics: Using Mass Spectrometry To detect foodborne protein contaminants, as exemplified by shiga toxin variants and prion strains. J. Agric. Food Chem. 2018, 66, 8435–8450. [Google Scholar] [CrossRef] [PubMed]
- Tonacini, J.; Stephan, D.; Vogel, G.; Avondet, M.-A.; Kalman, F.; Crovadore, J.; Lefort, F.; Schnyder, B. Intact Staphylococcus Enterotoxin SEB from Culture Supernatant Detected by MALDI-TOF Mass Spectrometry. Toxins 2019, 11, 101. [Google Scholar] [CrossRef] [Green Version]
- Bittar, F.; Ouchenane, Z.; Smati, F.; Raoult, D.; Rolain, J.M. MALDI-TOF-MS for rapid detection of staphylococcal Panton-Valentine leukocidin. Int. J. Antimicrob. Agents 2009, 34, 467–470. [Google Scholar] [CrossRef] [Green Version]
- Gagnaire, J.; Dauwalder, O.; Boisset, S.; Khau, D.; Freydière, A.M.; Ader, F.; Bes, M.; Lina, G.; Tristan, A.; Reverdy, M.E.; et al. Detection of Staphylococcus aureus delta-toxin production by whole-cell MALDI-TOF mass spectrometry. PLoS ONE 2012, 7, e40660. [Google Scholar] [CrossRef]
- Nedelkov, D.; Rasooly, A.; Nelson, R.W. Multitoxin biosensor-mass spectrometry analysis: A new approach for rapid, real-time, sensitive analysis of staphylococcal toxins in food. Int. J. Food Microbiol. 2000, 60, 1–13. [Google Scholar] [CrossRef]
- Carrera, M.; Böhme, K.; Gallardo, J.M.; Barros-Velázquez, J.; Cañas, B.; Calo-Mata, P. Characterization of Foodborne Strains of Staphylococcus aureus by Shotgun Proteomics: Functional Networks, Virulence Factors and Species-Specific Peptide Biomarkers. Front. Microbiol. 2017, 8, 2458. [Google Scholar] [CrossRef] [Green Version]
- Abril, A.G.; Carrera, M.; Böhme, K.; Barros-Velázquez, J.; Cañas, B.; Rama, J.L.R.; Villa, T.G.; Calo-Mata, P. Characterization of Bacteriophage Peptides of Pathogenic Streptococcus by LC-ESI-MS/MS: Bacteriophage Phylogenomics and Their Relationship to Their Host. Front. Microbiol. 2020, 11, 1241. [Google Scholar] [CrossRef]
- Abril, A.G.; Carrera, M.; Böhme, K.; Barros-Velázquez, J.; Rama, J.L.R.; Calo-Mata, P.; Sánchez-Pérez, A.; Villa, T.G. Proteomic characterization of antibiotic resistance, and production of antimicrobial and virulence factors in Streptococcus species associated with bovine mastitis. Could enzybiotics represent novel therapeutic agents against these pathogens? Antibiotics 2020, 9, 302. [Google Scholar] [CrossRef]
- Egyir, B.; Hadjirin, N.F.; Gupta, S.; Owusu, F.; Agbodzi, B.; Adogla-Bessa, T.; Addo, K.K.; Stegger, M.; Larsen, A.R.; Holmes, M.A. Whole genome sequence profiling of antibiotic resistant Staphylococcus aureus isolates from livestock and farm attendants in Ghana. J. Glob. Antimicrob. Resist. 2020, 22, 527–532. [Google Scholar] [CrossRef] [PubMed]
- Chang, B.S.; Moon, J.S.; Kang, H.M.; Kim, Y.I.; Lee, H.K.; Kim, J.D.; Lee, B.S.; Koo, H.C.; Park, Y.H. Protective effects of recombinant staphylococcal enterotoxin type C mutant vaccine against experimental bovine infection by a strain of Staphylococcus aureus isolated from subclinical mastitis in dairy cattle. Vaccine 2008, 26, 2081–2091. [Google Scholar] [CrossRef] [PubMed]
- Karauzum, H.; Adhikari, R.P.; Sarwar, J.; Devi, V.S.; Abaandou, L.; Haudenschild, C.; Mahmoudieh, M.; Boroun, A.R.; Vu, H.; Nguyen, T.; et al. Structurally Designed Attenuated Subunit Vaccines for S. aureus LukS-PV and LukF-PV Confer Protection in a Mouse Bacteremia Model. PLoS ONE 2013, 8, e65384. [Google Scholar] [CrossRef] [PubMed]

| Toxin | Principal Toxins in Food Poisoning | Gene | Activity |
|---|---|---|---|
| Toxic shock syndrome toxin 1 (TSST-1) | TSST-1 | tst | superantigen activity |
| Staphylococcal enterotoxins (SE) | SEA, SEB, SEC1, SEC2, SEC3, to SEE. SEG to SER, and SEU | sea to see, seg to ser, and seu | superantigen activity |
| SE-like toxins | SEG, SEH, SEI, SER, SES, SEIY, and SET | seg, seh, sei, ser, ses, seiy, and set | superantigen activity. Without emetic properties or have not been tested yet |
| Leukocidins | Panton-Valentine leukocidin (PVL), LukPQ, LukMF’, LukAB, and LukED | lukPV, lukPQ, lukM, lukA and lukB genes, and lukED | pore-forming toxins |
| Hemolysins | α hemolysin (Hla) and β hemolysin (Hlb) | hla and hlb. | pore-forming toxins |
| Exfoliative toxins (ETs) | ETA to ETE | eta to ete | serine proteases that specifically cleave Dsg1 * |
| Test Kit | Manufacturer | Foods Covered | Features |
|---|---|---|---|
| VIDAS Staph enterotoxin (SET) Immunoassay | BioMérieux | Dairy products, meat, and seafood, etc. | N/A |
| VIDAS Staph enterotoxin II (SET2) Immunoassay | BioMérieux | Milk and milk products, canned foods, dehydrated foods, meat, seafood, and shellfish, etc. | Detects SEA-SEE in 80 min, providing accurate results within the same day. |
| TECRA Staphylococcal Enterotoxin VIA | 3M Microbiology Canned | Canned mushrooms, nonfat dry milk, canned lobster bisque, beef, and pasta, cooked chicken, and cheese | Fast, reliably detects SEA, SEB, SEC1, SEC2, SEC3, SED, and SEE from food, food-related products, and enrichment cultures with a sensitivity of 1 ng/mL |
| RIDASCREEN Immunoassay | R-Biopharm, Darmstadt, Germany | Various, including cheese | Sandwich enzyme immunoassay for the identification of SETs A, B, C, D, E in fluids and solid foods, as well as in bacterial cultures |
| Transia (Transiatube and TransiaPlate) Immunoaffinity, ELISA | Diffchamb, Lyon, France | Milk and dairy products | N/A |
| SET-RPLA | Oxoid | A wide variety of food and food products such as dairy, meat, and meat products | Detects staphylococcal enterotoxins in a wide variety of foods, providing a semiquantitative result. Sensitivity of the test is 1 ng/mL of extract |
| SET-RPLA “SEIKEN” RPLA | Denka Seiken | RPLA test employing separately sensitized with highly specific antibodies for SEs A, B, C, and D. Results are semiquantitative |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
G. Abril, A.; G. Villa, T.; Barros-Velázquez, J.; Cañas, B.; Sánchez-Pérez, A.; Calo-Mata, P.; Carrera, M. Staphylococcus aureus Exotoxins and Their Detection in the Dairy Industry and Mastitis. Toxins 2020, 12, 537. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins12090537
G. Abril A, G. Villa T, Barros-Velázquez J, Cañas B, Sánchez-Pérez A, Calo-Mata P, Carrera M. Staphylococcus aureus Exotoxins and Their Detection in the Dairy Industry and Mastitis. Toxins. 2020; 12(9):537. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins12090537
Chicago/Turabian StyleG. Abril, Ana, Tomás G. Villa, Jorge Barros-Velázquez, Benito Cañas, Angeles Sánchez-Pérez, Pilar Calo-Mata, and Mónica Carrera. 2020. "Staphylococcus aureus Exotoxins and Their Detection in the Dairy Industry and Mastitis" Toxins 12, no. 9: 537. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins12090537





