Evaluating the Pollution Risk of Soil Due to Natural Drainage of Orange Peel: First Results
Abstract
:1. Introduction
2. Materials and Methods
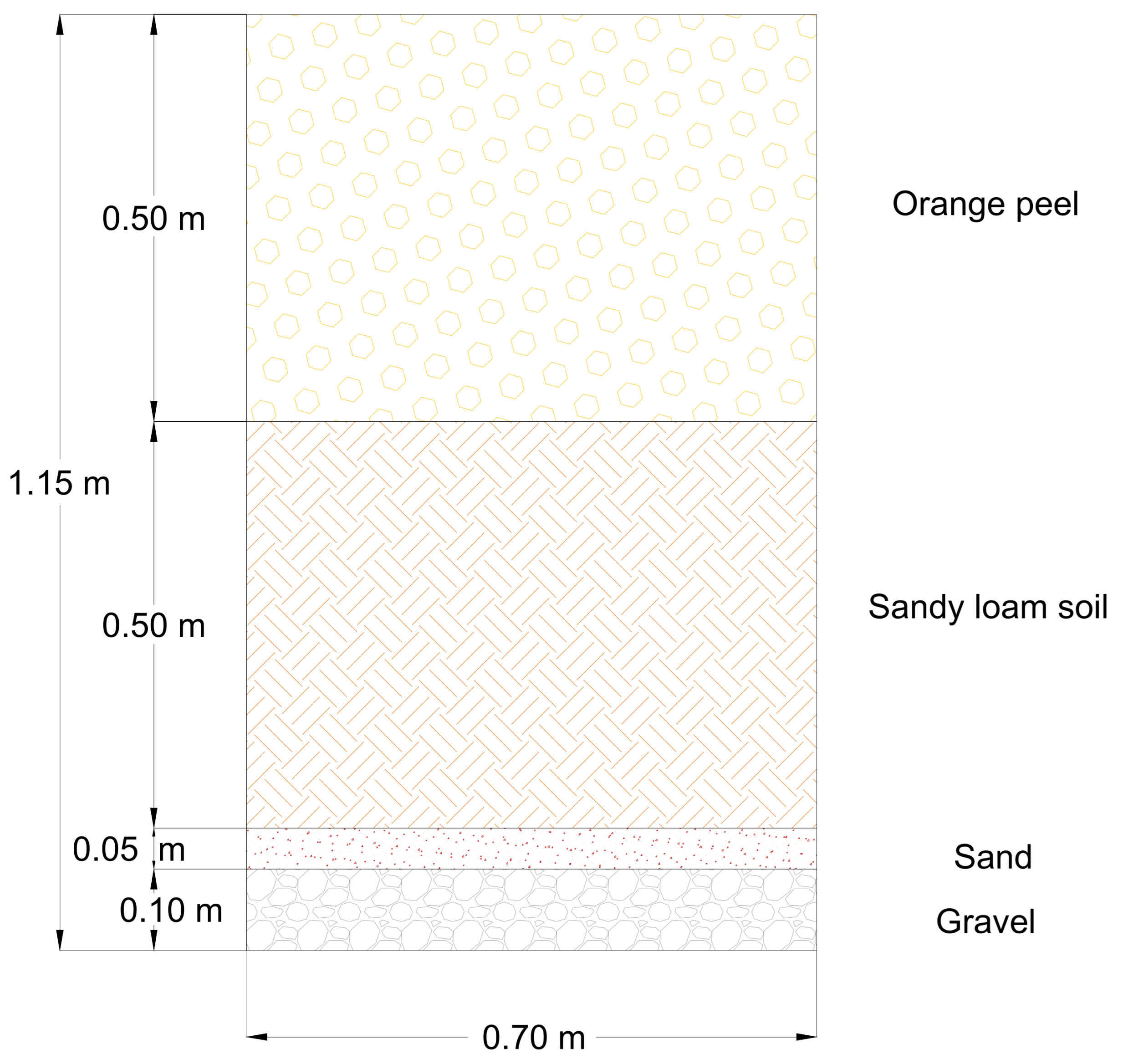
2.1. Description of the Experimental Tests
- Raw OP (“R” test);
- Already naturally-drained OP (“ND” test), then exposed on day 39 to a simulated rainfall of 100 mm (equivalent to about 38.5 L and to 20% v/v of the OP volume);
- Already solar-dried OP (“SD” test), then exposed to the same simulated rainfall.
2.2. Soil, OP and Leachate Characterisation
3. Results and Discussion
- A slight increase in pH in naturally-drained (“ND” test, pH = 7.8) and solar-dried (“SD”, 7.9) OP compared to the raw biomass (“R”, 7.3);
- A noticeably higher concentration of COD in the leachate released from raw OP (“R” test, COD = 4.4 g L−1) compared to the OP exposed to the rainfall (“ND”, 1.8 g L−1 and “SD”, 0.7 g L−1);
- A total COD amount in the leachate from the raw OP (“R” test) of 166 g, that is about 20-fold and 50-fold higher than that released from the biomass subjected to rainfall simulation (“ND”, 8.8 g and “SD”, 3.1 g).
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Zema, D.A.; Calabrò, P.S.; Folino, A.; Tamburino, V.; Zappia, G.; Zimbone, S.M. Valorisation of citrus processing waste: A review. Waste Manag. 2018, 80, 252–273. [Google Scholar] [CrossRef] [PubMed]
- FAO FAOSTAT—Crops Production. Available online: http://www.fao.org/faostat/en/#data (accessed on 4 May 2021).
- Nam, I.S.; Garnsworthy, P.C.; Ahn, J.H. Supplementation of essential oil extracted from citrus peel to animal feeds decreases microbial activity and aflatoxin contamination without disrupting in vitro ruminal fermentation. Asian Australasian J. Anim. Sci. 2006, 19, 1617–1622. [Google Scholar] [CrossRef]
- Bousbia, N.; Abert, M.; Ferhat, M.A.; Meklati, B.Y.; Chemat, F. A new process for extraction of essential oil from Citrus peels: Microwave hydrodiffusion and gravity. J. Food Eng. 2009, 90, 409–413. [Google Scholar] [CrossRef]
- Mahato, N.; Sharma, K.; Koteswararao, R.; Sinha, M.; Baral, E.; Cho, M.H. Citrus essential oils: Extraction, authentication and application in food preservation. Crit. Rev. Food Sci. Nutr. 2017, 4, 1–15. [Google Scholar] [CrossRef]
- Hull, W.Q.; Lindsay, C.W.; Baier, W.E. Chemicals from oranges. Ind. Eng. Chem. 1953, 45, 876–890. [Google Scholar] [CrossRef]
- Moufida, S.; Marzouk, B. Biochemical characterization of blood orange, sweet orange, lemon, bergamot and bitter orange. Phytochemistry 2003, 62, 1283–1289. [Google Scholar] [CrossRef]
- Arce, A.; Marchiaro, A.; Martínez-Ageitos, J.M.; Soto, A. Citrus Essential Oil Deterpenation by Liquid-Liquid Extraction. Can. J. Chem. Eng. 2005, 83, 366–370. [Google Scholar] [CrossRef]
- Zema, D.A.; Calabro, P.S.; Folino, A.; Tamburino, V.; Zappia, G.; Zimbone, S.M. Wastewater management in citrus processing industries: An overview of advantages and limits. Water 2019, 11, 2481. [Google Scholar] [CrossRef] [Green Version]
- Goodrich, R.M.; Braddock, R.J. Major By-Products of the Florida Citrus Processing Industry; University of Florida: Gainesville, FL, USA, 2006. [Google Scholar]
- Kimball, D.A. Citrus Processing: A Complete Guide; Springer: New York, NY, USA, 1999; ISBN 9781461549734. [Google Scholar]
- Sharma, K.; Mahato, N.; Cho, M.H. Converting citrus wastes into value-added products: Economic and environmently friendly approaches. Nutrition 2017, 34, 29–46. [Google Scholar] [CrossRef] [PubMed]
- Calabrò, P.S.; Pontoni, L.; Porqueddu, I.; Greco, R.; Pirozzi, F.; Malpei, F. Effect of the concentration of essential oil on orange peel waste biomethanization: Preliminary batch results. Waste Manag. 2016, 48, 440–447. [Google Scholar] [CrossRef]
- Zema, D.A. Planning the optimal site, size, and feed of biogas plants in agricultural districs. Biofuels Bioprod. Biorefining 2017, 11, 454–471. [Google Scholar] [CrossRef]
- John, I.; Muthukumar, K.; Arunagiri, A. A review on the potential of citrus waste for D-Limonene, pectin, and bioethanol production. Int. J. Green Energy 2017, 14, 599–612. [Google Scholar] [CrossRef]
- Satari, B.; Palhed, J.; Karimi, K.; Lundin, M.; Taherzadeh, M.J.; Zamani, A. Process optimization for citrus waste biorefinery via simultaneous pectin extraction and pretreatment. BioResources 2017, 12, 1706–1722. [Google Scholar] [CrossRef] [Green Version]
- Gelsomino, A.; Abenavoli, M.R.; Princi, G.; Attinà, E.; Cacco, G.; Sorgonà, A. Compost from Fresh Orange Waste: A Suitable Substrate for Nursery and Field Crops? Compost Sci. Util. 2010, 18, 201–210. [Google Scholar] [CrossRef]
- Sorgonà, A.; Abenavoli, M.R.; Cacco, G.; Gelsomino, A. Growth of Tomato and Zucchini Seedlings in Orange Waste Compost Media: pH and Implication of Dosage. Compost Sci. Util. 2011, 19, 189–196. [Google Scholar] [CrossRef]
- Gelsomino, A.; Abenavoli, M.R.; Sorgonà, A. Above- and below-ground morphological responses of a citrus rootstock interfered with orange waste compost: An evaluation as a component of growing media. Agrochimica 2014, 58, 148–164. [Google Scholar]
- Bampidis, V.A.; Robinson, P.H. Citrus by-products as ruminant feeds: A review. Anim. Feed Sci. Technol. 2006, 128, 175–217. [Google Scholar] [CrossRef]
- Madrid, J.; Hernández, F.; Pulgar, M.A.; Cid, J.M. Urea and citrus by-product supplementation of straw-based diets for goats: Effect on barley straw digestibility. Small Rumin. Res. 1997, 24, 149–155. [Google Scholar] [CrossRef]
- Gohl, B.I. Citru by-product for animal feed. In Ruminant Nutrition: Selected Articles from the World Animal Review, FAO Animal Production and Halth Paper; FAO: Rome, Italy, 1978. [Google Scholar]
- Caparra, P.; Foti, F.; Scerra, M.; Sinatra, M.C.; Scerra, V. Solar-dried citrus pulp as an alternative energy source in lamb diets: Effects on growth and carcass and meat quality. Small Rumin. Res. 2007, 68, 303–311. [Google Scholar] [CrossRef]
- Tamburino, V.; Zema, D.A.; Zimbone, S.M. Orange Peel Utilizations in Southern Italy. In Proceedings of the CIGR Section VI International Symposium on Food and Agricultural Products: Processing and Innovations, Naples, Italy, 24–26 September 2007. [Google Scholar]
- Bombino, G.; Denisi, P.; Fortugno, D.; Tamburino, V.; Zema, D.A.; Zimbone, S.M. Land spreading of solar-dried citrus peel to control runoff and soil erosion. WIT Trans. Ecol. Environ. 2010, 140, 145–154. [Google Scholar]
- Górka, A. Threats to Rural Landscape and Its Protection in Poland. Environments 2018, 5, 109. [Google Scholar] [CrossRef] [Green Version]
- Huang, T.; Lo, K. Effects of Land Use Change on Sediment and Water Yields in Yang Ming Shan National Park, Taiwan. Environments 2015, 2, 32–42. [Google Scholar] [CrossRef] [Green Version]
- Diodato, N.; Guerriero, L.; Bellocchi, G. Modeling and Upscaling Plot-Scale Soil Erosion under Mediterranean Climate Variability. Environments 2017, 4, 58. [Google Scholar] [CrossRef] [Green Version]
- Botta, G.; Brignoli, V.; Alberti, M.; Riva, G.; Scrosta, V.; Toscano, G. Analisi delle iniziative per la produzione di energia elettrica da biomasse agro-industriali in Italia. In Proceedings of the Atti IV Convegno Nazionale “Utilizzazione Termica dei Rifiuti”—Relazioni Tecniche, Abano Terme, Italy, 12–13 June 2003; pp. 12–13. [Google Scholar]
- Siles, J.Á.; Martín, M.D.L.Á.; Martín, A.; Raposo, F.; Borja, R. Anaerobic digestion of wastewater derived from the pressing of orange peel generated in orange juice production. J. Agric. Food Chem. 2007, 55, 1905–1914. [Google Scholar] [CrossRef] [PubMed]
- Negro, V.; Mancini, G.; Ruggeri, B.; Fino, D. Citrus waste as feedstock for bio-based products recovery: Review on limonene case study and energy valorization. Bioresour. Technol. 2016, 214, 806–815. [Google Scholar] [CrossRef]
- Tamburino, V.; Zimbone, S.M. Solar drying processes of citrus peel. In Proceedings of the XIII International CIGR Congress on Agricultural Engineering, Rabat, Marocco, 2–6 February 1997; pp. 2–6. [Google Scholar]
- Andiloro, S.; Tamburino, V.; Zimbone, S.M. Indagini sui processi di drenaggio ed evaporazione da pastazzo di arancia. In Valorizzazione di Acque Reflue e Sottoprodotti dell’Industria Agrumaria e Olearia; Laruffa Editore: Reggio Calabria, Italy, 2004. [Google Scholar]
- Fazzino, F.; Mauriello, F.; Paone, E.; Sidari, R.; Calabrò, P.S. Integral valorization of orange peel waste through optimized ensiling: Lactic acid and bioethanol production. Chemosphere 2021, 271, 129602. [Google Scholar] [CrossRef]
- Calabrò, P.S.; Panzera, M.F. Biomethane production tests on ensiled orange peel waste. Int. J. Heat Technol. 2017, 35, 130–136. [Google Scholar] [CrossRef]
- TT Le, A.; Kasahara, T.; Vudhivanich, V. Seasonal Variation and Retention of Ammonium in Small Agricultural Streams in Central Thailand. Environments 2018, 5, 78. [Google Scholar] [CrossRef] [Green Version]
- Malini, R.; Tajuddin, R.M.; Sa’adah, N.; Hamid, A. Citrus Fruit Waste Leachate Treatment By Using Newly Developed Flat Sheet Membrane. Int. J. Sci. Res. Publ. 2018, 8, 83. [Google Scholar] [CrossRef]
- Bombino, G.; Denisi, P.; Gómez, J.; Zema, D. Water Infiltration and Surface Runoff in Steep Clayey Soils of Olive Groves under Different Management Practices. Water 2019, 11, 240. [Google Scholar] [CrossRef] [Green Version]
- Bombino, G.; Denisi, P.; Gómez, J.A.; Zema, D.A. Mulching as best management practice to reduce surface runoff and erosion in steep clayey olive groves. Int. Soil Water Conserv. Res. 2021, 9, 26–36. [Google Scholar] [CrossRef]
- Walkley, A.; Black, I.A. An examination of the degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Sci. 1934, 37, 29–38. [Google Scholar] [CrossRef]
- Bremmer, J.; Mulvaney, C. Nitrogen total. In Methods of Soil Analysis; Page, A., Miller, R., Keeney, D., Eds.; American Society of Agronomy: Madison, WI, USA, 1982; pp. 595–624. [Google Scholar]
- IRSA-CNR. Metodi Analitici per le Acque; IRSA-CNR, Ed.; Istituto di Ricerca Sulle Acque, Consiglio Nazionale delle Ricerche: Rome, Italy, 1994. [Google Scholar]


| Parameter | Method | Measuring Unit | Value |
|---|---|---|---|
| pH | - | - | 9.0 |
| Organic matter | Walkey and Black method, 1934 [40] | % weight | 1.5 |
| Total nitrogen | Bremmer and Mulvaney method, 1982 [41] | 1.4 |
| Parameter | Instrument or Method | Measuring Unit | Value |
|---|---|---|---|
| pH | pH-meter, Hanna Instruments HI98230 | - | 7.3 |
| Dry matter * | Oven drying at 105 °C until weight stabilisation | % | 15.6 |
| Electrical conductivity | Portable conductivimeter, Hanna Instruments HI933000 | mS cm−1 | 1.7 |
| Parameter | Measuring Unit | OP Test | ||
|---|---|---|---|---|
| R | ND | SD | ||
| pH | - | 7.3 | 7.8 | 7.9 |
| Electrical conductivity | mS cm−1 | 1.4 | 2.2 | 1.2 |
| Cumulated volume | L | 37.7 | 4.9 | 4.4 |
| Chemical Oxygen Demand (COD) | g L−1 | 4.4 | 1.8 | 0.7 |
| Total COD | g | 166 | 8.8 | 3.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andiloro, S.; Calabrò, P.S.; Folino, A.; Zema, D.A.; Zimbone, S.M. Evaluating the Pollution Risk of Soil Due to Natural Drainage of Orange Peel: First Results. Environments 2021, 8, 43. https://0-doi-org.brum.beds.ac.uk/10.3390/environments8050043
Andiloro S, Calabrò PS, Folino A, Zema DA, Zimbone SM. Evaluating the Pollution Risk of Soil Due to Natural Drainage of Orange Peel: First Results. Environments. 2021; 8(5):43. https://0-doi-org.brum.beds.ac.uk/10.3390/environments8050043
Chicago/Turabian StyleAndiloro, Serafina, Paolo Salvatore Calabrò, Adele Folino, Demetrio Antonio Zema, and Santo Marcello Zimbone. 2021. "Evaluating the Pollution Risk of Soil Due to Natural Drainage of Orange Peel: First Results" Environments 8, no. 5: 43. https://0-doi-org.brum.beds.ac.uk/10.3390/environments8050043







