Ketone- and Cyano-Selenoesters to Overcome Efflux Pump, Quorum-Sensing, and Biofilm-Mediated Resistance
Abstract
:1. Introduction
2. Results
2.1. Determination of Minimum Inhibitory Concentrations by Microdilution Method
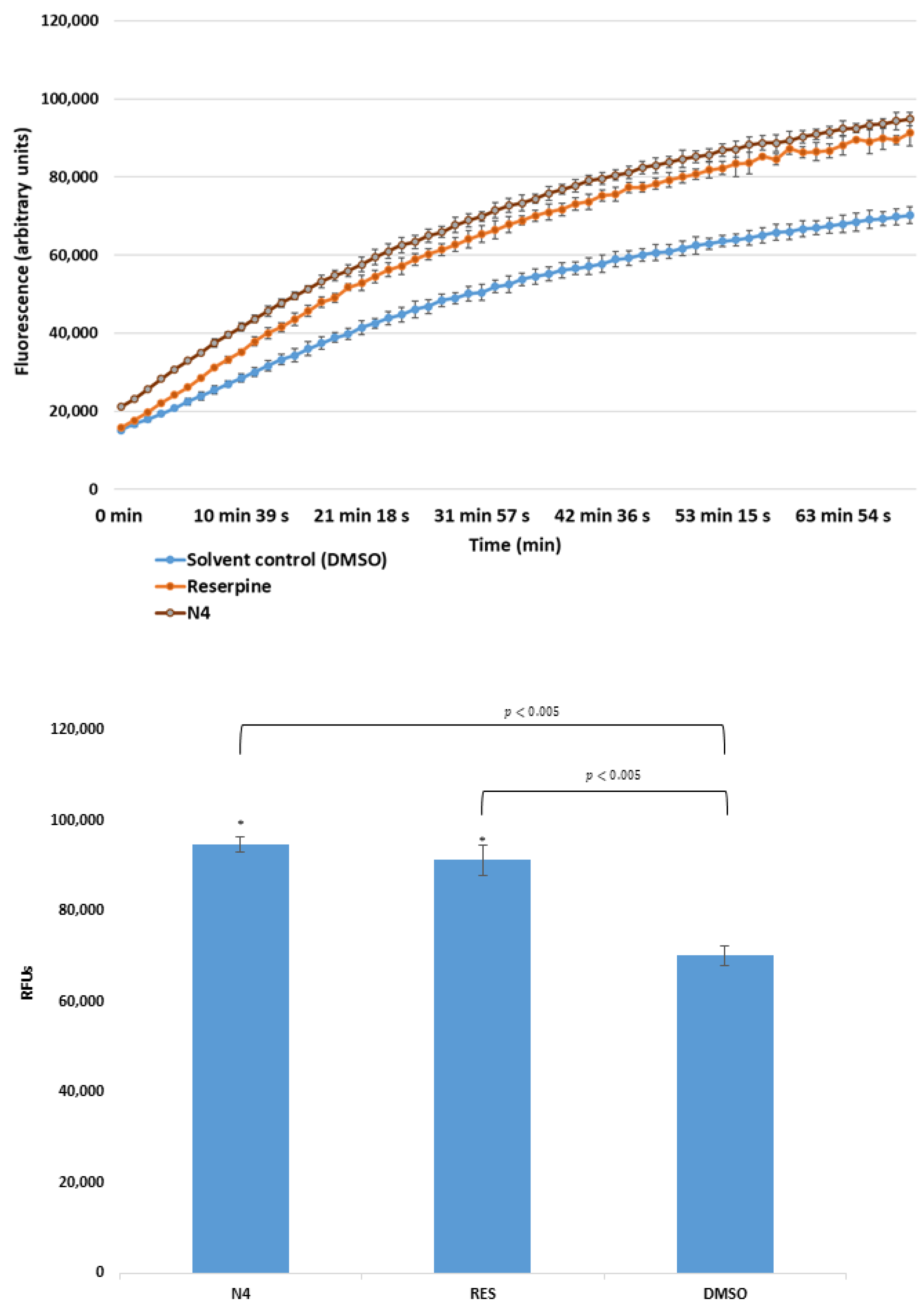
2.2. Real-Time Ethidium Bromide Accumulation Assay
2.3. Assay for Quorum Sensing (QS) Inhibition
2.4. Anti-Biofilm Activity
3. Discussion
3.1. Antibacterial Activity
3.2. Efflux Pump Inhibitory Asssay
3.3. Quorum Sensing (QS) Inhibition and Anti-Biofilm Assay
4. Materials and Methods
4.1. Compounds
4.2. Reagents and Media
4.3. Bacterial Strains
4.4. Determination of Minimum Inhibitory Concentrations (MIC) by Microdilution Method
4.5. Real-Time Ethidium Bromide Accumulation Assay
4.6. Assay for Quorum Sensing (QS) Inhibition
4.7. Anti-Biofilm Activity
4.7.1. Inhibition of Biofilm Formation
4.7.2. Disruption of Mature Biofilm
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ventola, C.L. The antibiotic resistance crisis: part 1: Causes and threats. P T Peer-Rev. J. Formul. Manag. 2015, 40, 277. [Google Scholar]
- Bragg, R.R.; Meyburgh, C.M.; Lee, J.-Y.; Coetzee, M. Potential Treatment Options in a Post-antibiotic Era. Adv. Exp. Med. Biol. 2018, 1052, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Coyne, L.A.; Latham, S.M.; Dawson, S.; Donald, I.J.; Pearson, R.B.; Smith, R.F.; Williams, N.J.; Pinchbeck, G.L. Exploring Perspectives on Antimicrobial Use in Livestock: A Mixed-Methods Study of UK Pig Farmers. Front. Vet. Sci. 2019, 6, 257. [Google Scholar] [CrossRef] [Green Version]
- Dong, L.T.; Espinoza, H.V.; Espinoza, J.L. Emerging superbugs: The threat of Carbapenem Resistant Enterobacteriaceae. AIMS Microbiol. 2020, 7, 176–182. [Google Scholar] [CrossRef] [PubMed]
- El-Hamid, M.I.A.; El-Naenaeey, E.-S.Y.; Kandeel, T.M.; Hegazy, W.A.H.; Mosbah, R.A.; Nassar, M.S.; Bakhrebah, M.A.; Abdulaal, W.H.; Alhakamy, N.A.; Bendary, M.M. Promising Antibiofilm Agents: Recent Breakthrough against Biofilm Producing Methicillin-Resistant Staphylococcus aureus. Antibiot 2020, 9, 667. [Google Scholar] [CrossRef]
- Singh, S.; Singh, S.K.; Chowdhury, I.; Singh, R. Understanding the Mechanism of Bacterial Biofilms Resistance to Antimicrobial Agents. Open Microbiol. J. 2017, 11, 53–62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Costerton, J.W.; Stewart, P.S.; Greenberg, E.P. Bacterial Biofilms: A Common Cause of Persistent Infections. Science 1999, 284, 1318–1322. [Google Scholar] [CrossRef] [Green Version]
- Davey, M.E.; O’Toole, G.A. Microbial Biofilms: from Ecology to Molecular Genetics. Microbiol. Mol. Biol. Rev. 2000, 64, 847–867. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.-H.; Tian, X. Quorum Sensing and Bacterial Social Interactions in Biofilms. Sensors 2012, 12, 2519–2538. [Google Scholar] [CrossRef]
- Miller, M.B.; Bassler, B.L. Quorum Sensing in Bacteria. Annu. Rev. Microbiol. 2001, 55, 165–199. [Google Scholar] [CrossRef] [Green Version]
- Alcalde-Rico, M.; Hernando-Amado, S.; Blanco, P.; Martínez, J. Multidrug Efflux Pumps at the Crossroad between Antibiotic Resistance and Bacterial Virulence. Front. Microbiol. 2016, 7, 1483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yazhiniprabha, M.; Vaseeharan, B. In vitro and in vivo toxicity assessment of selenium nanoparticles with significant larvicidal and bacteriostatic properties. Mater. Sci. Eng. C 2019, 103, 109763. [Google Scholar] [CrossRef] [PubMed]
- Cihalova, K.; Chudobova, D.; Michalek, P.; Moulick, A.; Guran, R.; Kopel, P.; Adam, V.; Kizek, R. Staphylococcus aureus and MRSA Growth and Biofilm Formation after Treatment with Antibiotics and SeNPs. Int. J. Mol. Sci. 2015, 16, 24656–24672. [Google Scholar] [CrossRef] [PubMed]
- Jastrzebska, I.; Mellea, S.; Salerno, V.; Grzes, P.A.; Siergiejczyk, L.; Niemirowicz-Laskowska, K.; Bucki, R.; Monti, B.; Santi, C.; Laskowska, N.-. PhSeZnCl in the Synthesis of Steroidal β-Hydroxy-Phenylselenides Having Antibacterial Activity. Int. J. Mol. Sci. 2019, 20, 2121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ngo, H.X.; Shrestha, S.K.; Green, K.D.; Garneau-Tsodikova, S. Development of ebsulfur analogues as potent antibacterials against methicillin-resistant Staphylococcus aureus. Bioorganic Med. Chem. 2016, 24, 6298–6306. [Google Scholar] [CrossRef] [Green Version]
- Gajdács, M.; Spengler, G.; Sanmartín, C.; Marć, M.A.; Handzlik, J.; Domínguez-Álvarez, E. Selenoesters and selenoanhydrides as novel multidrug resistance reversing agents: A confirmation study in a colon cancer MDR cell line. Bioorganic Med. Chem. Lett. 2017, 27, 797–802. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kharma, A.; Misak, A.; Grman, M.; Brezova, V.; Kurakova, L.; Barath, P.; Jacob, C.; Chovanec, M.; Ondrias, K.; Domínguez-Álvarez, E. Release of reactive selenium species from phthalic selenoanhydride in the presence of hydrogen sulfide and glutathione with implications for cancer research. New J. Chem. 2019, 43, 11771–11783. [Google Scholar] [CrossRef] [Green Version]
- Reich, H.J.; Hondal, R.J. Why Nature Chose Selenium. ACS Chem. Biol. 2016, 11, 821–841. [Google Scholar] [CrossRef]
- Fernandez-Lazaro, D.; Fernandez-Lazaro, C.I.; Mielgo-Ayuso, J.; Navascués, L.J.; Córdova, A.; Seco-Calvo, J. The Role of Selenium Mineral Trace Element in Exercise: Antioxidant Defense System, Muscle Performance, Hormone Response, and Athletic Performance. A Systematic Review. Nutrients 2020, 12, 1790. [Google Scholar] [CrossRef]
- Stolwijk, J.M.; Garje, R.; Sieren, J.C.; Buettner, G.R.; Zakharia, Y. Understanding the Redox Biology of Selenium in the Search of Targeted Cancer Therapies. Antioxidants 2020, 9, 420. [Google Scholar] [CrossRef]
- Sumner, S.E.; Markley, R.L.; Kirimanjeswara, G.S. Role of Selenoproteins in Bacterial Pathogenesis. Biol. Trace Element Res. 2019, 192, 69–82. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P. The importance of selenium to human health. Lancet 2000, 356, 233–241. [Google Scholar] [CrossRef] [Green Version]
- Mosolygó, T.; Kincses, A.; Mosolygó, T.; Marć, M.A.; Nové, M.; Gajdács, M.; Sanmartín, C.; McNeil, H.E.; Blair, J.M.A.; Domínguez-Álvarez, E. Antiviral, Antimicrobial and Antibiofilm Activity of Selenoesters and Selenoanhydrides. Molecules 2019, 24, 4264. [Google Scholar] [CrossRef] [Green Version]
- Nové, M.; Kincses, A.; Szalontai, B.; Rácz, B.; Blair, J.M.A.; González-Prádena, A.; Benito-Lama, M.; Domínguez-Álvarez, E.; Spengler, G. Biofilm Eradication by Symmetrical Selenoesters for Food-Borne Pathogens. Microorganisms 2020, 8, 566. [Google Scholar] [CrossRef] [Green Version]
- Mosolygó, T.; Kincses, A.; Csonka, A.; Tönki, Á.S.; Witek, K.; Sanmartín, C.; Marć, M.A.; Handzlik, J.; Kieć-Kononowicz, K.; Domínguez-Álvarez, E.; et al. Selenocompounds as Novel Antibacterial Agents and Bacterial Efflux Pump Inhibitors. Molecules 2019, 24, 1487. [Google Scholar] [CrossRef] [Green Version]
- Peña-Morán, O.A.; Villarreal, M.L.; Alvarez, L.; Meneses-Acosta, A.; Rodríguez-López, V. Cytotoxicity, Post-Treatment Recovery, and Selectivity Analysis of Naturally Occurring Podophyllotoxins from Bursera fagaroides var. fagaroides on Breast Cancer Cell Lines. Molecules 2016, 21, 1013. [Google Scholar] [CrossRef]
- Spengler, G.; Kincses, A.; Gajdács, M.; Amaral, L. New Roads Leading to Old Destinations: Efflux Pumps as Targets to Reverse Multidrug Resistance in Bacteria. Molecules 2017, 22, 468. [Google Scholar] [CrossRef] [Green Version]
- Yong, Y.-C.; Zhong, J.-J. Impacts of Quorum Sensing on Microbial Metabolism and Human Health. In Future Trends in Biotechnology; Zhong, J.-J., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; Volume 131, pp. 25–61. [Google Scholar] [CrossRef]
- Chatterjee, R.; Shreenivas, M.M.; Sunil, R.; Chakravortty, D. Enteropathogens: Tuning Their Gene Expression for Hassle-Free Survival. Front. Microbiol. 2019, 9, 3303. [Google Scholar] [CrossRef]
- Gibbons, S.; Oluwatuyi, M.; Kaatz, G.W. A novel inhibitor of multidrug efflux pumps in Staphylococcus aureus. J. Antimicrob. Chemother. 2003, 51, 13–17. [Google Scholar] [CrossRef] [Green Version]
- Rezzonico, F.; Smits, T.H.M.; Duffy, B. Detection of AI-2 Receptors in Genomes of Enterobacteriaceae Suggests a Role of Type-2 Quorum Sensing in Closed Ecosystems. Sensors 2012, 12, 6645–6665. [Google Scholar] [CrossRef] [Green Version]
- Taga, M.E.; Semmelhack, J.L.; Bassler, B.L. The LuxS-dependent autoinducer AI-2 controls the expression of an ABC transporter that functions in AI-2 uptake in Salmonella typhimurium. Mol. Microbiol. 2008, 42, 777–793. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Domínguez Álvarez, E.; Spengler, G.; Jacob, C.; Sanmartín Grijalba, M.C. Selenoester-Containing Compounds for Use in the Treatment of Microbial Infections or Colorectal Cancer. European Patent EP18382693, 28 September 2018. [Google Scholar]
- Christopher, P.J.; Polgar, E.P. (Eds.) Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically, 11th ed.; Clinical and Laboratory Standards Institute: Wayne, MI, USA, 2018. [Google Scholar]
- Viktorova, J.; Stupák, M.; Rehorova, K.; Dobiasova, S.; Hoang, L.; Hajslova, J.; Van Thanh, T.; Van Tri, L.; Van Tuan, N.; Ruml, T. Lemon Grass Essential Oil does not Modulate Cancer Cells Multidrug Resistance by Citral—Its Dominant and Strongly Antimicrobial Compound. Foods 2020, 9, 585. [Google Scholar] [CrossRef] [PubMed]
- Hoang, L.; Beneš, F.; Fenclova, M.; Kronusova, O.; Švarcová, V.; Rehorova, K.; Švecová, E.B.; Vosátka, M.; Hajslova, J.; Kaštánek, P.; et al. Phytochemical Composition and In Vitro Biological Activity of Iris spp. (Iridaceae): A New Source of Bioactive Constituents for the Inhibition of Oral Bacterial Biofilms. Antibiot 2020, 9, 403. [Google Scholar] [CrossRef] [PubMed]
- Sandberg, M.E.; Schellmann, D.; Brunhofer, G.; Erker, T.; Busygin, I.; Leino, R.; Vuorela, P.; Fallarero, A. Pros and cons of using resazurin staining for quantification of viable Staphylococcus aureus biofilms in a screening assay. J. Microbiol. Methods 2009, 78, 104–106. [Google Scholar] [CrossRef] [PubMed]
- Haney, E.F.; Trimble, M.J.; Cheng, J.T.; Vallé, Q.; Hancock, R.E.W. Critical Assessment of Methods to Quantify Biofilm Growth and Evaluate Antibiofilm Activity of Host Defence Peptides. Biomolecules 2018, 8, 29. [Google Scholar] [CrossRef] [Green Version]



| Cpds. | RSe | R1 | R2 | R3 | Ring | Cpds. | RSe | R1 | R2 | R3 | Ring |
|---|---|---|---|---|---|---|---|---|---|---|---|
| K1 | -CH2COCH3 | -H | -H | -H | Thiophene | N1 | -CH2CN | -H | -H | -H | Thiophene |
| K2 | -CH2COCH3 | 2-F | -H | -H | Benzene | N2 | -CH2CN | 3-F | -H | -H | Benzene |
| K3 | -CH2COCH3 | 4-Br | -H | -H | Benzene | N3 | -CH2CN | 4-Br | -H | -H | Benzene |
| K4 | -CH2COCH3 | 2-CF3 | -H | -H | Benzene | N4 | -CH2CN | 2-CF3 | -H | -H | Benzene |
| K5 | -CH2COCH3 | 3-CF3 | -H | -H | Benzene | N5 | -CH2CN | 3-CF3 | -H | -H | Benzene |
| K6 | -CH2COCH3 | 3-Cl | 4-F | -H | Benzene | N6 | -CH2CN | 3-Cl | 4-F | -H | Benzene |
| K7 | -CH2COCH3 | 4-C(CH)3 | -H | -H | Benzene | N7 | -CH2CN | 3-CF3 | 5-CF3 | -H | Benzene |
| K8 | -CH2COCH3 | 2-F | 4-F | 5-F | Benzene |
| MIC Determination (µM) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Cpds. | S. aureus ATCC 25923 | S. aureus MRSA ATCC 43300 | S. aureus MRSA 272123 | S. Typhimurium SE01 Wild-Type | S. Typhimurium SE02 ΔacrB | S. Typhimurium SE03 ΔacrA | S. Typhimurium SE39 ΔtolC | P. aeruginosa CCM 3955 | P. aeruginosa NEM 986 |
| K1 | 1.56 | 1.56 | 0.78 | 50 | 100 | 100 | 100 | 100 | 50 |
| K2 | 1.56 | 3.125 | 0.78 | >100 | >100 | >100 | >100 | 100 | 50 |
| K3 | 1.56 | 3.125 | 0.78 | 50 | 50 | 50 | 50 | >100 | >100 |
| K4 | 3.125 | 3.125 | 1.56 | >100 | >100 | >100 | >100 | 100 | 50 |
| K5 | 1.56 | 3.125 | 0.78 | 100 | 50 | 50 | >100 | 100 | 50 |
| K6 | 1.56 | 3.125 | 0.39 | 100 | 100 | 100 | 100 | 100 | 50 |
| K7 | 1.56 | 1.56 | 0.39 | 50 | >100 | 100 | >100 | 100 | 100 |
| K8 | 1.56 | 1.56 | 0.78 | 50 | >100 | 100 | 100 | 100 | 50 |
| N1 | 12.5 | 100 | 25 | 50 | 50 | 100 | 100 | >100 | >100 |
| N2 | 12.5 | 100 | 50 | 50 | 100 | 100 | 100 | >100 | >100 |
| N3 | 12.5 | 50 | 25 | 50 | 50 | 50 | 50 | >100 | >100 |
| N4 | 12.5 | 100 | 50 | 100 | 100 | 100 | 100 | >100 | >100 |
| N5 | 12.5 | 50 | 50 | 100 | 100 | 100 | 100 | >100 | >100 |
| N6 | 12.5 | 50 | 25 | 100 | 50 | 100 | 100 | >100 | >100 |
| N7 | 12.5 | 50 | 25 | 50 | 50 | 50 | 100 | >100 | >100 |
| Relative Fluorescence Index (RFI) | ||||||
|---|---|---|---|---|---|---|
| Cpds. | S. aureus MRSA ATCC 43300 | S.aureus ATCC 25923 | S. Typhimurium SE01 wild-type | S. Typhimurium SE02 ΔacrB | S. Typhimurium SE03 ΔacrA | S. Typhimurium SE39 ΔtolC |
| K1 | −0.02 | 1.19 | 0.17 | 0.20 | 0.29 | 0.31 |
| K2 | −0.04 | 1.12 | 0.18 | 0.21 | 0.30 | 0.42 |
| K3 | −0.10 | 1.17 | 0.60 | 0.27 | 0.24 | 0.44 |
| K4 | −0.09 | 1.02 | 0.19 | 0.35 | 0.68 | 0.95 |
| K5 | −0.08 | 1.17 | 0.41 | 0.11 | 0.35 | 0.91 |
| K6 | −0.09 | 1.19 | 0.17 | 0.43 | 0.52 | 0.80 |
| K7 | −0.02 | 1.13 | 1.02 | 0.30 | 1.15 | 1.67 |
| K8 | −0.04 | 1.10 | 0.08 | 0.31 | 0.28 | 0.70 |
| N1 | 0.05 | 1.40 | 0.04 | 0.10 | 0.37 | 0.84 |
| N2 | −0.02 | 1.31 | −0.06 | −0.05 | 0.16 | 0.19 |
| N3 | −0.05 | 1.49 | 0.003 | 0.03 | 0.38 | 0.24 |
| N4 | 0.35 | 1.78 | 0.22 | 0.39 | 0.36 | 1.00 |
| N5 | −0.03 | 1.43 | 0.02 | 0.02 | 0.32 | 0.43 |
| N6 | −0.01 | 1.32 | 0.03 | −0.03 | 0.45 | 0.38 |
| N7 | −0.06 | 0.28 | 0.003 | 0.14 | 0.32 | 1.11 |
| CCCP | - | - | 3.37 | 1.83 | 3.30 | 1.61 |
| RES | 0.30 | 5.5 | - | - | - | - |
| Cpd. | Vibrio campbellii BAA 1118 | Vibrio campbellii BAA 1119 | ||||
|---|---|---|---|---|---|---|
| IC50 (µM) | EC50 (µM) | SI | IC50 (µM) | EC50 (µM) | SI | |
| K1 | 5.76 ± 0.07 | 0.22 ± 0.01 | 26.2 | 2.02 ± 0.15 | 0.71 ± 0.05 | 2.8 |
| K2 | 4.38 ± 0.47 | 0.25 ± 0.03 | 17.5 | 3.23 ± 0.14 | 0.22 ± 0.02 | 14.7 |
| K3 | 1.12 ± 0.02 | 0.17 ± 0.01 | 6.6 | 0.77 ± 0.07 | 0.23 ± 0.02 | 3.3 |
| K4 | 33.18 ± 3.45 | 4.68 ± 0.32 | 7.1 | 6.66 ± 0.13 | 0.29 ± 0.05 | 23.0 |
| K5 | 2.42 ± 0.29 | 1.35 ± 0.03 | 1.8 | 1.32 ± 0.10 | 0.45 ± 0.01 | 2.9 |
| K6 | 3.28 ± 0.19 | 2.29 ± 0.02 | 1.4 | 0.97 ± 0.09 | 1.20 ± 0.00 | 0.8 |
| K7 | 10.54 ± 0.19 | 1.77 ± 0.19 | 6.0 | 4.27 ± 0.23 | 0.15 ± 0.01 | 28.5 |
| K8 | 1.23 ± 0.07 | 0.11 ± 0.01 | 11.2 | 1.39 ± 0.03 | 0.46 ± 0.03 | 3.0 |
| N1 | 2.21 ± 0.19 | 1.45 ± 0.02 | 1.5 | 2.28 ± 0.12 | 0.26 ± 0.03 | 8.8 |
| N2 | 7.36 ± 0.70 | 0.34 ± 0.04 | 21.6 | 2.40 ± 0.12 | 0.73 ± 0.02 | 5.1 |
| N3 | 2.199 ± 0.16 | 0.34 ± 0.04 | 6.5 | 2.35 ± 0.03 | <0.06 | 37.6 |
| N4 | 2.52 ± 0.03 | 1.29 ± 0.04 | 2.0 | 6.41 ± 0.42 | 0.73 ± 0.02 | 8.8 |
| N5 | 12.51 ± 0.05 | >5 | - | 3.57 ± 0.08 | >5 | - |
| N6 | 1.37 ± 0.02 | 0.37 ± 0.05 | 3.7 | 2.28 ± 0.12 | 0.22 ± 0.00 | 10.4 |
| N7 | 3.84 ± 0.15 | 1.44 ± 0.06 | 2.7 | 7.71 ± 0.10 | 0.25 ± 0.02 | 30.8 |
| Compounds | Staphylococcus aureus ATCC 25923 | Pseudomonas aeruginosa CCM 3955 | ||
|---|---|---|---|---|
| Anti-Adhesion (μM) | Anti-Biofilm (μM) | Anti-Adhesion (μM) | Anti-Biofilm (μM) | |
| K1 | 1.84 ± 0.26 | 32.80 ± 3.25 | 1.15 ± 0.01 | 10.21 ± 0.48 |
| K2 | 1.72 ± 0.17 | 28.08 ± 1.17 | 1.10 ± 0.11 | 8.78 ± 0.66 |
| K3 | 1.39 ± 0.13 | 11.64 ± 0.99 | 1.14 ± 0.05 | 6.00 ± 0.74 |
| K4 | 3.59 ± 0.48 | 28.70 ± 4.18 | 3.04 ± 0.33 | 21.85 ± 2.04 |
| K5 | 2.84 ± 0.13 | 15.44 ± 0.42 | 1.51 ± 0.22 | 6.45 ± 0.30 |
| K6 | 2.96 ± 0.16 | 12.87 ± 0.37 | 2.33 ± 0.25 | 14.29 ± 1.62 |
| K7 | 3.08 ± 0.24 | 40.80 ± 3.12 | 2.16 ± 0.29 | 11.06 ± 1.92 |
| K8 | 1.35 ± 0.16 | 9.22 ± 0.61 | 0.86 ± 0.09 | 6.98 ± 0.22 |
| N1 | 2.46 ± 0.15 | 24.79 ± 2.65 | 1.78 ± 0.07 | 15.51 ± 1.65 |
| N2 | 3.14 ± 0.12 | 48.08 ± 3.82 | 2.86 ± 0.17 | 18.06 ± 0.72 |
| N3 | 1.19 ± 0.15 | 30.46 ± 2.72 | 0.92 ± 0.01 | 10.56 ± 0.95 |
| N4 | 1.49 ± 0.08 | 28.91 ± 2.00 | 2.49 ± 0.43 | 13.48 ± 0.82 |
| N5 | 3.01 ± 0.35 | 34.55 ± 3.00 | 3.40 ± 0.10 | 24.81 ± 2.12 |
| N6 | 1.83 ± 0.15 | 21.75 ± 2.61 | 1.34 ± 0.08 | 13.46 ± 1.77 |
| N7 | 1.99 ± 0.26 | 16.53 ± 0.76 | 1.81 ± 0.04 | 11.09 ± 0.82 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szemerédi, N.; Kincses, A.; Rehorova, K.; Hoang, L.; Salardón-Jiménez, N.; Sevilla-Hernández, C.; Viktorová, J.; Domínguez-Álvarez, E.; Spengler, G. Ketone- and Cyano-Selenoesters to Overcome Efflux Pump, Quorum-Sensing, and Biofilm-Mediated Resistance. Antibiotics 2020, 9, 896. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics9120896
Szemerédi N, Kincses A, Rehorova K, Hoang L, Salardón-Jiménez N, Sevilla-Hernández C, Viktorová J, Domínguez-Álvarez E, Spengler G. Ketone- and Cyano-Selenoesters to Overcome Efflux Pump, Quorum-Sensing, and Biofilm-Mediated Resistance. Antibiotics. 2020; 9(12):896. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics9120896
Chicago/Turabian StyleSzemerédi, Nikoletta, Annamária Kincses, Katerina Rehorova, Lan Hoang, Noemi Salardón-Jiménez, Clotilde Sevilla-Hernández, Jitka Viktorová, Enrique Domínguez-Álvarez, and Gabriella Spengler. 2020. "Ketone- and Cyano-Selenoesters to Overcome Efflux Pump, Quorum-Sensing, and Biofilm-Mediated Resistance" Antibiotics 9, no. 12: 896. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics9120896







