Known and Emerging Mycotoxins in Small- and Large-Scale Brewed Beer
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Reagents and Standards
2.3. Analysis for Mycotoxin Determination
2.4. Performances of the Methods
2.5. Statistical Analysis
3. Results and Discussion
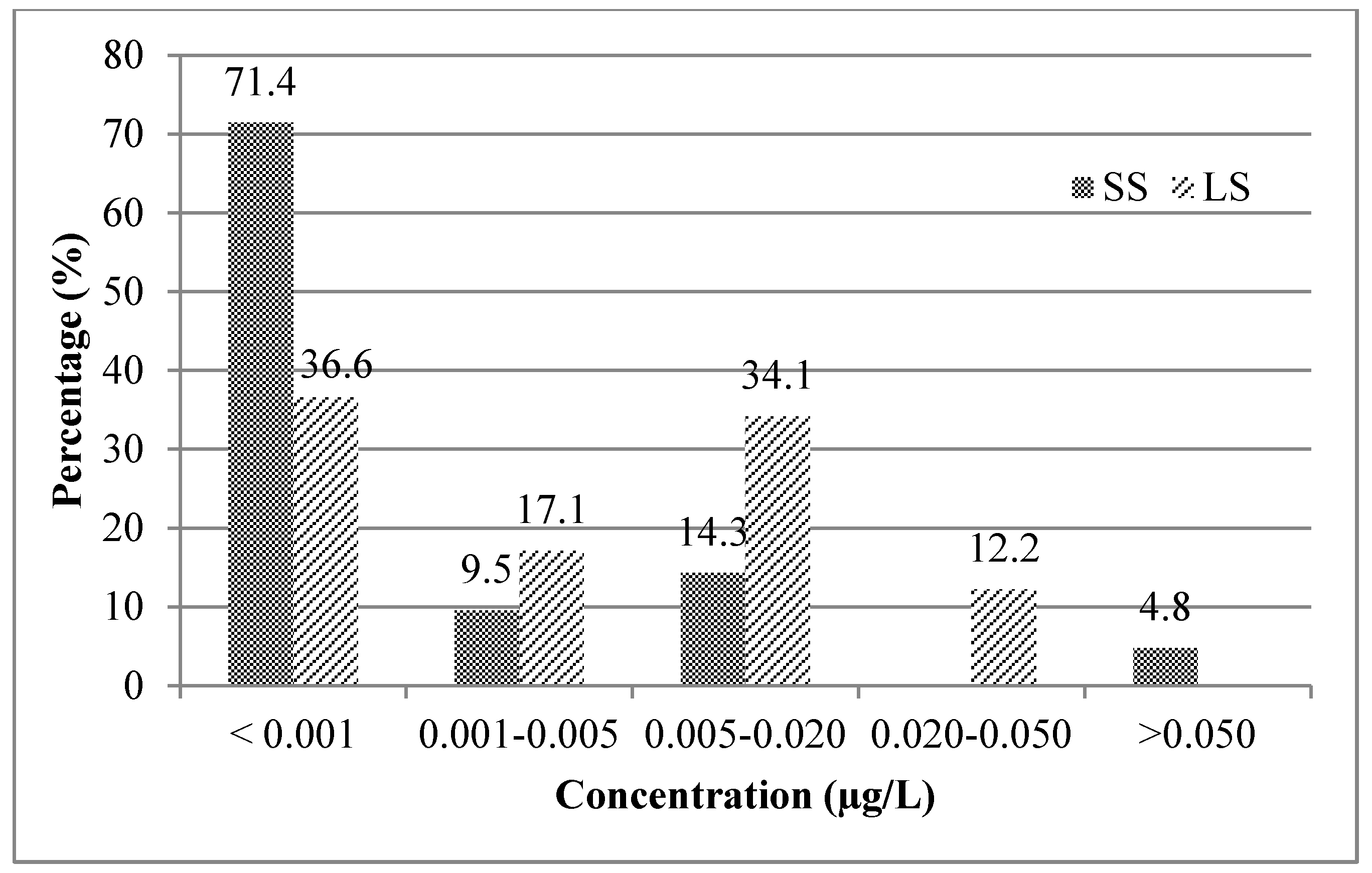
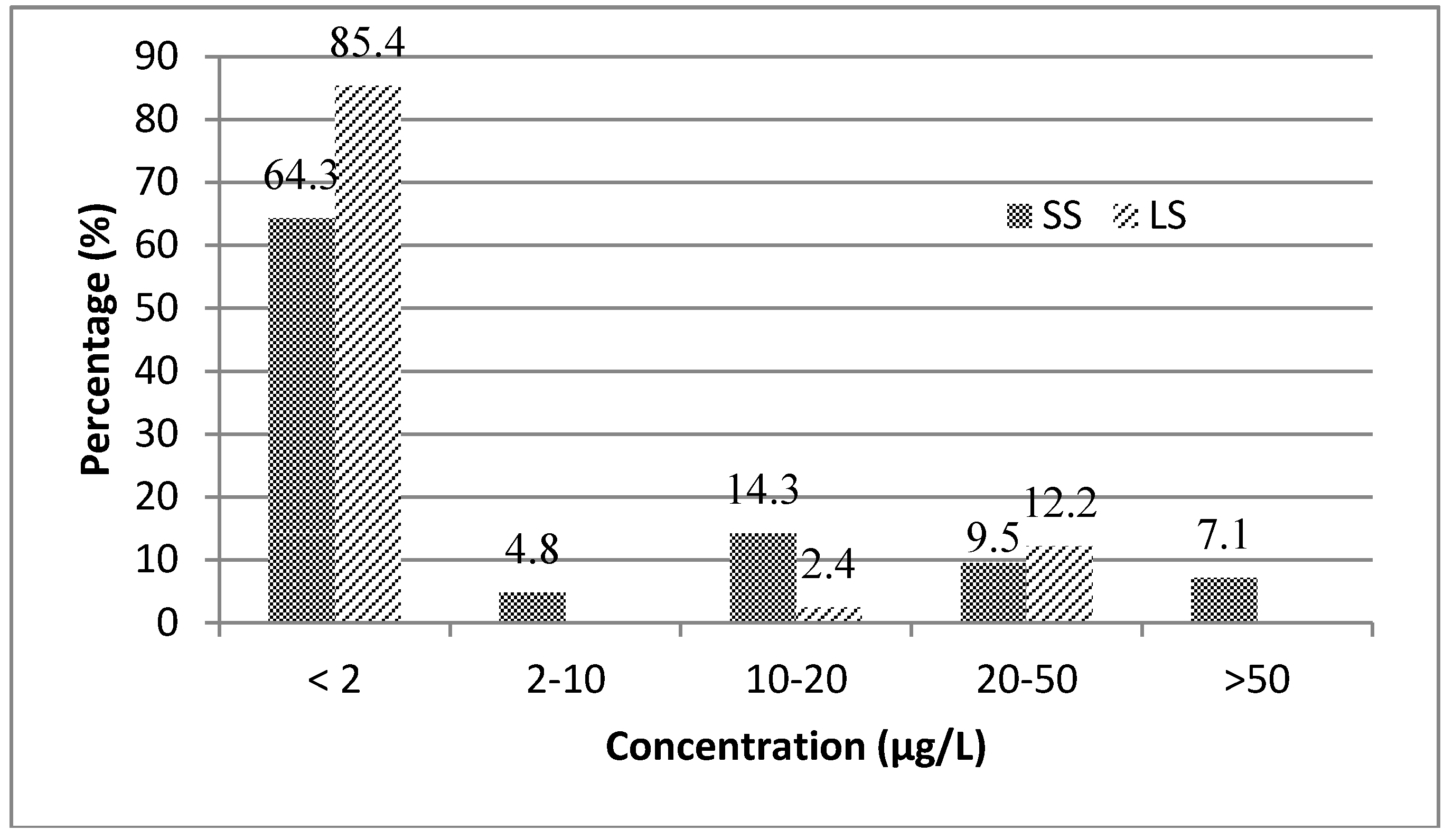
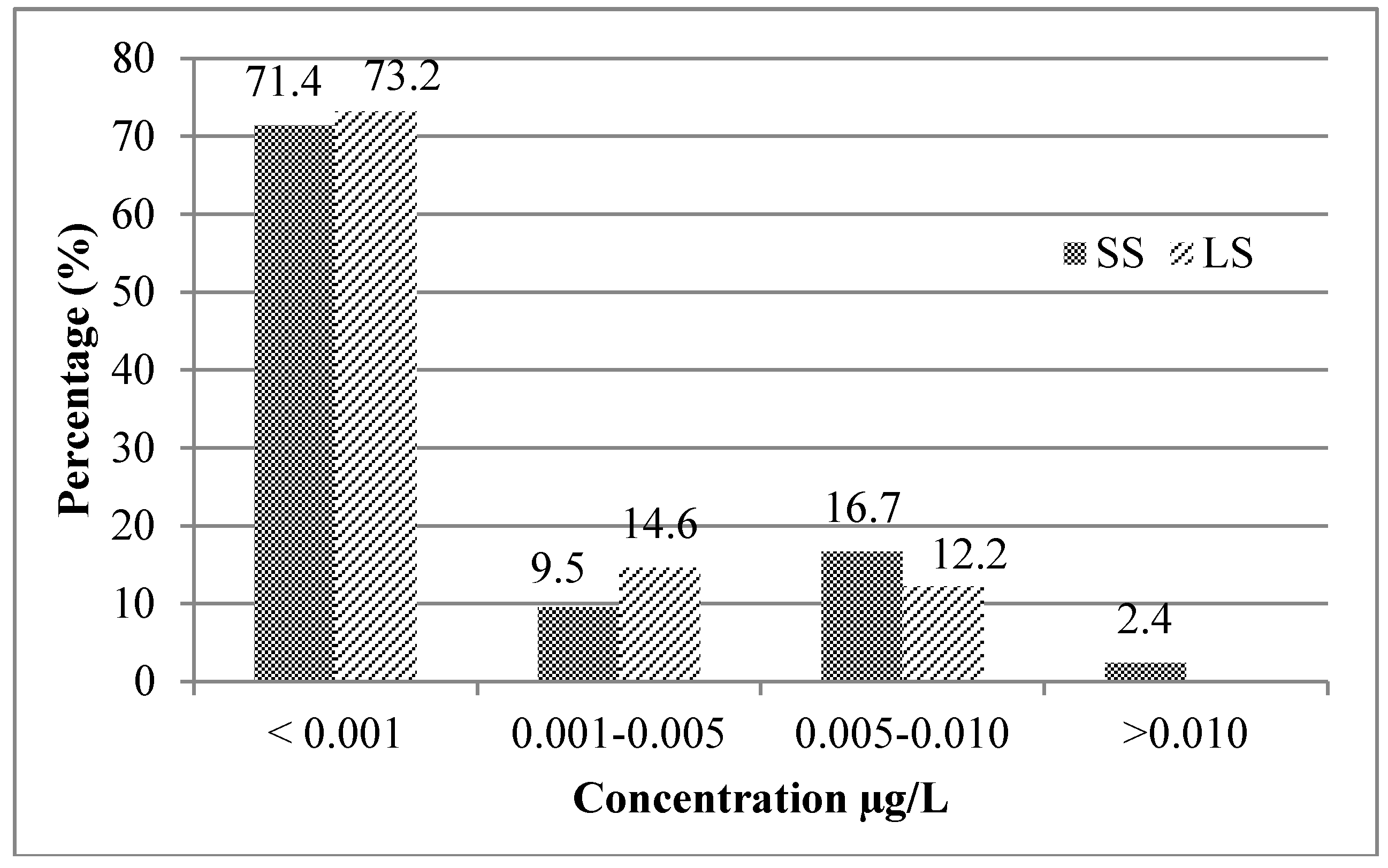
3.1. Mycotoxin Occurrence in Beer Samples
3.2. Mycotoxin Co-Occurrence
3.3. Estimation of Mycotoxin Dietary Intakes by Beer Consumption
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Baxter, E.D.; Slaiding, I.R.; Kelly, B. Behaviour of ochratoxin A in brewing. J. Am. Soc. Brew. Chem. 2001, 59, 98–100. [Google Scholar]
- Schwarz, P.B.; Casper, H.H.; Beattie, S. Fate and development of naturally occurring Fusarium mycotoxins during malting and brewing. J. Am. Soc. Brew. Chem. 1995, 53, 121–127. [Google Scholar]
- Mizutani, K.; Nagatomi, Y.; Mochizuki, N. Metabolism of zearalenone in the course of beer fermentation. Toxins 2011, 3, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Pietri, A.; Bertuzzi, T.; Agosti, B.; Donadini, G. Transfer of aflatoxin B1 and fumonisin B1 from naturally contaminated raw materials to beer during an industrial brewing process. Food Addit. Contam. 2010, 27, 1431–1439. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Scientific Opinion on the risks for public and animal health related to the presence of citrinin in food and feed. EFSA Panel on Contaminants in the Food Chain. EFSA J. 2012, 10, 2605. [Google Scholar] [CrossRef]
- EFSA CONTAM Panel (EFSA Panel on Contaminants in the Food Chain). Scientific Opinion on the risk for public and animal health related to the presence of sterigmatocystin in food and feed. EFSA J. 2013, 11, 3254. [Google Scholar] [CrossRef] [Green Version]
- International Agency for Research on Cancer. Some naturally occurring and synthetic food components, furocoumarins and ultraviolet radiation. In IARC Monographs on the Evaluation of Carcinogenic Risk to Humans; IARC, World Health Organization: Lyon, France, 1986; pp. 40–67. [Google Scholar]
- International Agency for Research on Cancer. Some naturally occurring substances: Food items and constituents, heterocyclic aromatic amines and mycotoxins. In IARC Monographs on the Evaluation of Carcinogenic Risk to Humans; IARC, World Health Organization: Lyon, France, 1993; Volume 56, pp. 245–395. [Google Scholar]
- International Agency for Research on Cancer. Aflatoxins IARC Monograph on the Evaluation of Carcinogenic Risks to Humans; IARC, World Health Organization: Lyon, France, 2002; Volume 82, pp. 171–300. [Google Scholar]
- Gazzetta Ufficiale della Repubblica Italiana (GU). Act of the Legislature, DDL S 1328-B, 28 July 2016 n° 154, GU 186 of 10 August 2016.
- Bertuzzi, T.; Rastelli, S.; Mulazzi, A.; Donadini, G.; Pietri, A. Mycotoxin occurrence in beer produced in several European countries. Food Control 2011, 22, 2059–2064. [Google Scholar] [CrossRef]
- Bertuzzi, T.; Romani, M.; Rastelli, S.; Mulazzi, A.; Pietri, A. Sterigmatocystin Occurrence in Paddy and Processed Rice Produced in Italy in the Years 2014–2015 and Distribution in Milled Rice Fractions. Toxins 2017, 9, 86. [Google Scholar] [CrossRef] [PubMed]
- Varga, J.; Kiss, R.; Matrai, T.; Teren, J. Detection of ochratoxin A in Hungarian wines and beers. Acta Aliment. 2005, 34, 381–392. [Google Scholar] [CrossRef]
- Medina, A.; Jimenez, M.; Gimeno Adelantado, J.V.; Valle Algarra, F.M.; Mateo, R. Determination of ochratoxin A in beer marketed in Spain by liquid chromatography with fluorescence detection using lead hydroxyacetate as a cleanup agent. J. Chromatogr. A 2005, 1083, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Lhotská, I.; Šatínský, D.; Havlíková, L.; Solich, P. A fully automated and fast method using direct sample injection combined with fused-core column on-line SPE–HPLC for determination of ochratoxin A and citrinin in lager beers. Anal. Bioanal. Chem. 2016, 408, 3319–3329. [Google Scholar] [CrossRef] [PubMed]
- Peters, J.; van Dam, R.; van Doorn, R.; Katerere, D.; Berthiller, F.; Haasnoot, W.; Nielen, M. Mycotoxin profiling of 1000 beer samples with a special focus on craft beer. PLoS ONE 2017, 12, e0185887. [Google Scholar] [CrossRef] [PubMed]
- Commission of the European Communities. Commission Regulation (EC) No 105/2010 of 5 February 2010 amending Regulation (EC) No 1881/2006 setting maximum levels for certain contaminants in foodstuffs as regards ochratoxin A. Off. J. Eur. Union 2010, L 35, 7–8. [Google Scholar]
- Papadopoulou-Bouraoui, A.; Vrabcheva, T.; Valzacchi, S.; Stroka, J.; Anklam, E. Screening survey of deoxynivalenol in beer from the European market by an enzyme-linked immunosorbent assay. Food Addit. Contam. 2004, 21, 607–617. [Google Scholar] [CrossRef] [PubMed]
- Anselme, M.; Tangni, E.; Pussemeier, L.; Motte, J.C.; Hove, F.; van Schneider, Y.J.; Peteghem, C.; van Larondelle, Y. Comparison of ochratoxin A and deoxynivalenol in organically and conventionally produced beers sold on the Belgiam market. Food Addit. Contam. 2006, 23, 910–918. [Google Scholar] [CrossRef] [PubMed]
- Kostelanska, M.; Hajslova, J.; Zachariasova, M.; Malachova, A.; Kalachova, K.; Poustka, J.; Fiala, J.; Scott, P.M.; Berthiller, F.; Krska, R. Occurrence of deoxynivalenol and its major conjugate, deoxynivalenol-3-glucoside, in beer and some brewing intermediates. J. Agric. Food Chem. 2009, 57, 3187–3194. [Google Scholar] [CrossRef] [PubMed]
- Bauer, J.; Gross, M.; Gottschalk, C.; Usleber, E. Investigations on the occurrence of mycotoxins in beer. Food Control 2016, 63, 135–139. [Google Scholar] [CrossRef]
- Piacentini, K.C.; Savi, G.D.; Olivo, G.; Scussel, V.M. Quality and occurrence of deoxynivalenol and fumonisins in craft beer. Food Control 2015, 50, 925–929. [Google Scholar] [CrossRef]
- Veršilovskis, A.; De Saeger, S.; Miķelsone, V. Determination of sterigmatocystin in beer by high performance liquid chromatography with ultraviolet detection. World Mycotoxin J. 2008, 1, 161–166. [Google Scholar] [CrossRef]
- Mol, H.; Mac Donald, S.; Anagnostopoulos, C.; Spanjer, M.; Bertuzzi, T.; Pietri, A. European survey on sterigmatocystin in cereals, cereals-based products, beer and nuts. World Mycotoxin J. 2016, 9, 633–642. [Google Scholar] [CrossRef]
- Baxter, E.D.; Byrd, N.; Slaiding, I.R. Food Safety Review of UK Cereal Grain for Use in Malting, Milling and Animal Feed; HGCA Project Report; Campden BRI: Gloucestershire, UK, 2009; pp. 464–522. [Google Scholar]
- Magan, N.; Aldred, D. Post-harvest control strategies: Minimizing mycotoxins in the food chain. Int. J. Food Microbiol. 2007, 119, 131–139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harcz, P.; Tangni, E.K.; Wilmart, O.; Moons, E.; Peteghem, C.; van Saeger, S.; de Schneider, Y.J.; Larondelle, Y.; Pussemeier, L. Intake of ochratoxin A and deoxynivalenol through beer consumption in Belgium. Food Addit. Contam. 2007, 24, 910–916. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gumus, T.; Arici, M.; Demirci, M. A survey of barley, malt and beer contamination with OTA in Turkey. J. Inst. Brew. 2004, 110, 146–149. [Google Scholar] [CrossRef]
| Beer | OTA (µg/L) | DON (µg/L) | STC (µg/L) | |
|---|---|---|---|---|
| Small-scale brewed beer | Positives (%) | 28.6 | 35.7 | 28.6 |
| Mean | 0.005 ± 0.014 | 11.3 ± 20.2 | 0.002 ± 0.003 | |
| Median | <LOD | <LOD | <LOD | |
| Maximum value | 0.070 | 99 | 0.018 | |
| Large-scale brewed beer | Positives (%) | 63.4 | 14.6 | 26.8 |
| Mean | 0.008 ± 0.011 | 5.8 ± 10.4 | 0.001 ± 0.002 | |
| Median | 0.0048 | <LOD | <LOD | |
| Maximum value | 0.045 | 46 | 0.009 |
| Mycotoxin | Daily Average Exposure (µg/kgbw) | Maximum Daily Exposure (µg/kgbw) | Tolerable Daily Intake (µg/kg bw) | % of Tolerable Daily Intake | |||
|---|---|---|---|---|---|---|---|
| SS beer | LS beer | SS beer | LS beer | Max level | |||
| OTA | 0.000007 | 0.000011 | 0.00010 | 0.017 | 0.04 | 0.067 | 0.59 |
| DON | 0.0162 | 0.0083 | 0.1419 | 1 | 1.62 | 0.83 | 14.2 |
| STC | 0.0000028 | 0.0000014 | 0.0000258 | / | / | / | / |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bertuzzi, T.; Rastelli, S.; Mulazzi, A.; Donadini, G.; Pietri, A. Known and Emerging Mycotoxins in Small- and Large-Scale Brewed Beer. Beverages 2018, 4, 46. https://0-doi-org.brum.beds.ac.uk/10.3390/beverages4020046
Bertuzzi T, Rastelli S, Mulazzi A, Donadini G, Pietri A. Known and Emerging Mycotoxins in Small- and Large-Scale Brewed Beer. Beverages. 2018; 4(2):46. https://0-doi-org.brum.beds.ac.uk/10.3390/beverages4020046
Chicago/Turabian StyleBertuzzi, Terenzio, Silvia Rastelli, Annalisa Mulazzi, Gianluca Donadini, and Amedeo Pietri. 2018. "Known and Emerging Mycotoxins in Small- and Large-Scale Brewed Beer" Beverages 4, no. 2: 46. https://0-doi-org.brum.beds.ac.uk/10.3390/beverages4020046





