Blood Mercury Levels in Children with Kawasaki Disease and Disease Outcome
Abstract
:1. Introduction
2. Materials and Methods
2.1. Collection of Blood Samples for Blood Mercury Estimations
2.2. Statistical Analyses
3. Results
3.1. Clinical Characteristics of Total KD Patients
3.2. Association of Mercury Levels with the Clinical Features and Outcomes of Kawasaki Disease
3.3. Association of Mercury Levels with the Laboratory Characteristics of Kawasaki Disease
3.4. Total Mercury Levels in Whole Blood for Different Populations
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chen, J.J.; Ma, X.J.; Liu, F.; Yan, W.L.; Huang, M.R.; Huang, M.; Huang, G.Y. Epidemiologic Features of Kawasaki Disease in Shanghai From 2008 Through 2012. Pediatr. Infect. Dis. J. 2016, 35, 7–12. [Google Scholar] [CrossRef]
- Rowley, A.H.; Shulman, S.T. The Epidemiology and Pathogenesis of Kawasaki Disease. Front. Pediatr. 2018, 6, 374. [Google Scholar] [CrossRef] [Green Version]
- Huang, Y.H.; Lin, K.M.; Ho, S.C.; Yan, J.H.; Lo, M.H.; Kuo, H.C. Increased Incidence of Kawasaki Disease in Taiwan in Recent Years: A 15 Years Nationwide Population-Based Cohort Study. Front. Pediatr. 2019, 7, 121. [Google Scholar] [CrossRef] [PubMed]
- Uehara, R.; Belay, E.D. Epidemiology of Kawasaki disease in Asia, Europe, and the United States. J. Epidemiol. 2012, 22, 79–85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yeter, D.; Portman, M.A.; Aschner, M.; Farina, M.; Chan, W.C.; Hsieh, K.S.; Kuo, H.C. Ethnic Kawasaki Disease Risk Associated with Blood Mercury and Cadmium in U.S. Children. Int. J. Environ. Res. Public Health 2016, 13, 101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yeter, D.; Deth, R. ITPKC susceptibility in Kawasaki syndrome as a sensitizing factor for autoimmunity and coronary arterial wall relaxation induced by thimerosal’s effects on calcium signaling via IP3. Autoimmun. Rev. 2012, 11, 903–908. [Google Scholar] [CrossRef] [PubMed]
- Pichichero, M.E.; Gentile, A.; Giglio, N.; Umido, V.; Clarkson, T.; Cernichiari, E.; Zareba, G.; Gotelli, C.; Gotelli, M.; Yan, L.; et al. Mercury levels in newborns and infants after receipt of thimerosal-containing vaccines. Pediatrics 2008, 121, e208–e214. [Google Scholar] [CrossRef] [Green Version]
- Committee on Infectious Diseases and Committee on Environmental Health; American Academy of Pediatrics. Thimerosal in vaccines—An interim report to clinicians. Pediatrics 1999, 104, 570–574. [Google Scholar]
- Kuo, H.C.; Hsu, Y.W.; Lo, M.H.; Huang, Y.H.; Chien, S.C.; Chang, W.C. Single-nucleotide polymorphism rs7251246 in ITPKC is associated with susceptibility and coronary artery lesions in Kawasaki disease. PLoS ONE 2014, 9, e91118. [Google Scholar] [CrossRef] [Green Version]
- Kuo, H.C.; Hsu, Y.W.; Wu, C.M.; Chen, S.H.; Hung, K.S.; Chang, W.P.; Yang, K.D.; Hsieh, K.S.; Chen, W.C.; Onouchi, Y.; et al. A replication study for association of ITPKC and CASP3 two-locus analysis in IVIG unresponsiveness and coronary artery lesion in Kawasaki disease. PLoS ONE 2013, 8, e69685. [Google Scholar] [CrossRef] [Green Version]
- Lou, J.; Xu, S.; Zou, L.; Zhong, R.; Zhang, T.; Sun, Y.; Lu, X.; Liu, L.; Li, C.; Wang, L.; et al. A functional polymorphism, rs28493229, in ITPKC and risk of Kawasaki disease: An integrated meta-analysis. Mol. Biol. Rep. 2012, 39, 11137–11144. [Google Scholar] [CrossRef] [PubMed]
- Yeter, D.; Deth, R.; Kuo, H.C. Mercury promotes catecholamines which potentiate mercurial autoimmunity and vasodilation: Implications for inositol 1,4,5-triphosphate 3-kinase C susceptibility in kawasaki syndrome. Korean Circ. J. 2013, 43, 581–591. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.T.; Wang, J.K.; Yeh, J.I.; Sun, L.C.; Chen, P.L.; Wu, J.F.; Chang, C.C.; Lee, W.L.; Shen, C.T.; Wang, N.K.; et al. Clinical Implication of the C Allele of the ITPKC Gene SNP rs28493229 in Kawasaki Disease: Association With Disease Susceptibility and BCG Scar Reactivation. Pediatr. Infect. Dis. J. 2011, 30, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Singh, S. BCG Site Reactivation in Kawasaki Disease. Arthritis Rheumatol. 2016, 68, 2026. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gamez-Gonzalez, L.B.; Hamada, H.; Llamas-Guillen, B.A.; Ruiz-Fernandez, M.; Yamazaki-Nakashimada, M. BCG and Kawasaki disease in Mexico and Japan. Hum. Vaccines Immunother. 2017, 13, 1091–1093. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, C.C.; Chang, J.W.; Huang, H.Y.; Chen, H.L. Factors influencing blood mercury levels of inhabitants living near fishing areas. Sci. Total Environ. 2012, 424, 316–321. [Google Scholar] [CrossRef]
- You, S.H.; Wang, S.L.; Pan, W.H.; Chan, W.C.; Fan, A.M.; Lin, P. Risk assessment of methylmercury based on internal exposure and fish and seafood consumption estimates in Taiwanese children. Int. J. Hyg. Environ. Health 2018, 221, 697–703. [Google Scholar] [CrossRef]
- Park, J.D.; Zheng, W. Human exposure and health effects of inorganic and elemental mercury. J. Prev. Med. Public Health Yebang Uihakhoe Chi 2012, 45, 344–352. [Google Scholar] [CrossRef]
- Sharma, B.M.; Sanka, O.; Kalina, J.; Scheringer, M. An overview of worldwide and regional time trends in total mercury levels in human blood and breast milk from 1966 to 2015 and their associations with health effects. Environ. Int. 2019, 125, 300–319. [Google Scholar] [CrossRef]
- Chang, L.S.; Ming-Huey Guo, M.; Lo, M.H.; Kuo, H.C. Identification of increased expression of activating Fc receptors and novel findings regarding distinct IgE and IgM receptors in Kawasaki disease. Pediatr. Res. 2019. [Google Scholar] [CrossRef]
- Ling-Sai, C.; Guo, M.M.; Yan, J.H.; Huang, Y.H.; Lo, M.H.; Kuo, H.C. Low FCMR mRNA expression in leukocytes of patients with Kawasaki disease six months after disease onset. Pediatr. Allergy Immunol. 2020. [Google Scholar] [CrossRef] [PubMed]
- McCrindle, B.W.; Rowley, A.H.; Newburger, J.W.; Burns, J.C.; Bolger, A.F.; Gewitz, M.; Baker, A.L.; Jackson, M.A.; Takahashi, M.; Shah, P.B.; et al. Diagnosis, Treatment, and Long-Term Management of Kawasaki Disease: A Scientific Statement for Health Professionals From the American Heart Association. Circulation 2017, 135, e927–e999. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.S.; Hsu, Y.W.; Lu, C.C.; Lo, M.H.; Hsieh, K.S.; Li, S.C.; Chang, W.C.; Kuo, H.C. CYP2E1 Gene Polymorphisms Related to the Formation of Coronary Artery Lesions in Kawasaki Disease. Pediatr. Infect. Dis. J. 2017, 36, 1039–1043. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Hwang, M.S.; Ko, A.; Jeong, D.H.; Kang, H.S.; Yoon, H.J.; Hong, J.H. Total mercury concentrations in the general Korean population, 2008–2011. Regul. Toxicol. Pharmacol. RTP 2014, 70, 681–686. [Google Scholar] [CrossRef]
- Ilmiawati, C.; Yoshida, T.; Itoh, T.; Nakagi, Y.; Saijo, Y.; Sugioka, Y.; Sakamoto, M.; Ikegami, A.; Ogawa, M.; Kayama, F. Biomonitoring of mercury, cadmium, and lead exposure in Japanese children: A cross-sectional study. Environ. Health Prev. Med. 2015, 20, 18–27. [Google Scholar] [CrossRef] [Green Version]
- Jain, R.B. Trends in and factors affecting the observed levels of urinary inorganic and total blood mercury among US children, adolescents, adults, and senior citizens over 2005–2012. Environ. Toxicol. Pharmacol. 2017, 56, 268–281. [Google Scholar] [CrossRef]
- Gao, Z.Y.; Li, M.M.; Wang, J.; Yan, J.; Zhou, C.C.; Yan, C.H. Blood mercury concentration, fish consumption and anthropometry in Chinese children: A national study. Environ. Int. 2018, 110, 14–21. [Google Scholar] [CrossRef]
- Taylor, C. Range Rule for Standard Deviation. Available online: https://www.thoughtco.com/range-rule-for-standard-deviation-3126231 (accessed on 27 March 2020).
- Tu, S.H.; Hung, Y.T.; Chang, H.Y.; Hang, C.M.; Shaw, N.S.; Lin, W.; Lin, Y.C.; Hu, S.W.; Yang, Y.H.; Wu, T.C.; et al. Nutrition and Health Survey of Taiwan Elementary School Children 2001-2002: Research design, methods and scope. Asia Pac. J. Clin. Nutr. 2007, 16, 507–517. [Google Scholar]
- Schulz, C.; Wilhelm, M.; Heudorf, U.; Kolossa-Gehring, M. Update of the reference and HBM values derived by the German Human Biomonitoring Commission. Int. J. Hyg. Environ. Health 2011, 215, 26–35. [Google Scholar] [CrossRef]
- Ye, B.J.; Kim, B.G.; Jeon, M.J.; Kim, S.Y.; Kim, H.C.; Jang, T.W.; Chae, H.J.; Choi, W.J.; Ha, M.N.; Hong, Y.S. Evaluation of mercury exposure level, clinical diagnosis and treatment for mercury intoxication. Ann. Occup. Environ. Med. 2016, 28, 5. [Google Scholar] [CrossRef] [Green Version]
- Araki, T.; Kodera, A.; Kitada, K.; Fujiwara, M.; Muraoka, M.; Abe, Y.; Ikeda, M.; Tsukahara, H. Analysis of factors associated with development of Bacille Calmette-Guerin inoculation site change in patients with Kawasaki disease. J. Int. Med. Res. 2018, 46, 1640–1648. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carter, M.; Abdi, A.; Naz, F.; Thabet, F.; Vyas, A. A Mercury Toxicity Case Complicated by Hyponatremia and Abnormal Endocrinological Test Results. Pediatrics 2017, 140, e20161402. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, R.; Chansouria, J.P. Alterations in plasma sodium and potassium levels following chronic oral ingestion of lead, mercury and cadmium in male albino rats. Indian J. Exp. Biol. 1991, 29, 767–769. [Google Scholar] [PubMed]
- Brake, J.; Hester, P.Y.; Thaxton, P.; Morgan, G.W.; Sikes, C.V. Effect of mercury toxiocity on serum osmolality, plasma sodium and potassium, and serum proteins in the chicken. Arch. Environ. Contam. Toxicol. 1980, 9, 431–436. [Google Scholar] [CrossRef]
- Gungor, O.; Ozkaya, A.K.; Kirik, S.; Dalkiran, T.; Gungor, G.; Isikay, S.; Davutoglu, M.; Dilber, C. Acute Mercury Poisoning in a Group of School Children. Pediatr. Emerg. Care 2019, 35, 696–699. [Google Scholar] [CrossRef]
- Wang, X.; Mukherjee, B.; Park, S.K. Does Information on Blood Heavy Metals Improve Cardiovascular Mortality Prediction? J. Am. Heart Assoc. 2019, 8, e013571. [Google Scholar] [CrossRef]
- Kwaansa-Ansah, E.E.; Adimado, A.A.; Nriagu, J.O.; Basu, N. Comparison of Three Analytical Methods for the Quantitation of Mercury in Environmental Samples from the Volta Lake, Ghana. Bull. Environ. Contam. Toxicol. 2016, 97, 677–683. [Google Scholar] [CrossRef]
- Bulska, E.; Krata, A.; Kałabun, M.; Wojciechowski, M. On the use of certified reference materials for assuring the quality of results for the determination of mercury in environmental samples. Environ. Sci. Pollut. Res. Int. 2017, 24, 7889–7897. [Google Scholar] [CrossRef] [Green Version]
- Liu, T.Y.; Hung, Y.M.; Huang, W.C.; Wu, M.L.; Lin, S.L. Do people from Taiwan have higher heavy metal levels than those from Western countries? Singap. Med. J. 2017, 58, 267–271. [Google Scholar] [CrossRef] [Green Version]
- Lin, C.H.; Hsu, Y.T.; Yen, C.C.; Chen, H.H.; Tseng, C.J.; Lo, Y.K.; Chan, J.Y.H. Association between heavy metal levels and acute ischemic stroke. J. Biomed. Sci. 2018, 25, 49. [Google Scholar] [CrossRef]
- United Nations Environment Programme. Global Mercury Assessment 2018; UN Environment Programme Chemicals and Health Branch: Geneva, Switzerland, 2019. [Google Scholar]
- Lee, M.J.; Chou, M.C.; Chou, W.J.; Huang, C.W.; Kuo, H.C.; Lee, S.Y.; Wang, L.J. Heavy Metals’ Effect on Susceptibility to Attention-Deficit/Hyperactivity Disorder: Implication of Lead, Cadmium, and Antimony. Int. J. Environ. Res. Public Health 2018, 15, 1221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruggieri, F.; Majorani, C.; Domanico, F.; Alimonti, A. Mercury in Children: Current State on Exposure through Human Biomonitoring Studies. Int. J. Environ. Res. Public Health 2017, 14, 519. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hung, P.C.; Chang, S.H.; Ou-Yang, C.C.; Chang, M.B. Simultaneous removal of PCDD/Fs, pentachlorophenol and mercury from contaminated soil. Chemosphere 2016, 144, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.W.; Pai, M.C.; Chen, H.L.; Guo, H.R.; Su, H.J.; Lee, C.C. Cognitive function and blood methylmercury in adults living near a deserted chloralkali factory. Environ. Res. 2008, 108, 334–339. [Google Scholar] [CrossRef] [PubMed]
- Fang, G.C.; Wu, Y.S.; Chang, T.H. Comparison of atmospheric mercury (Hg) among Korea, Japan, China and Taiwan during 2000–2008. J. Hazard. Mater. 2009, 162, 607–615. [Google Scholar] [CrossRef]
| 1 Mercury Levels in Negative Presentation μg/L | 1 Mercury Levels in Positive Presentation μg/L | p-Value | |
|---|---|---|---|
| Changes in oral mucosa | 3.25 (0.95–8.33) | 3.00 (1.80–5.70) | 0.922 |
| Conjunctivitis | 3.00 (1.55–6.70) | 3.15 (1.78–5.75) | 0.884 |
| Palpable cervical lymphadenopathy | 3.60 (1.75–6.08) | 2.90 (1.80–5.30) | 0.448 |
| Edema of extremities | 3.20 (1.95–6.10) | 3.00 (1.70–5.80) | 0.947 |
| Skin rash | 3.40 (1.80–5.70) | 3.00 (1.75–5.95) | 0.825 |
| BCG 2 scar reactivation | 2.90 (1.60–5.30) | 3.40 (1.70–6.15) | 0.696 |
| Incomplete KD 3 | 2.90 (1.80–5.68) | 3.50 (1.50–6.40) | 0.900 |
| Variable | Blood Mercury | |
|---|---|---|
| Pearson r | p-Value | |
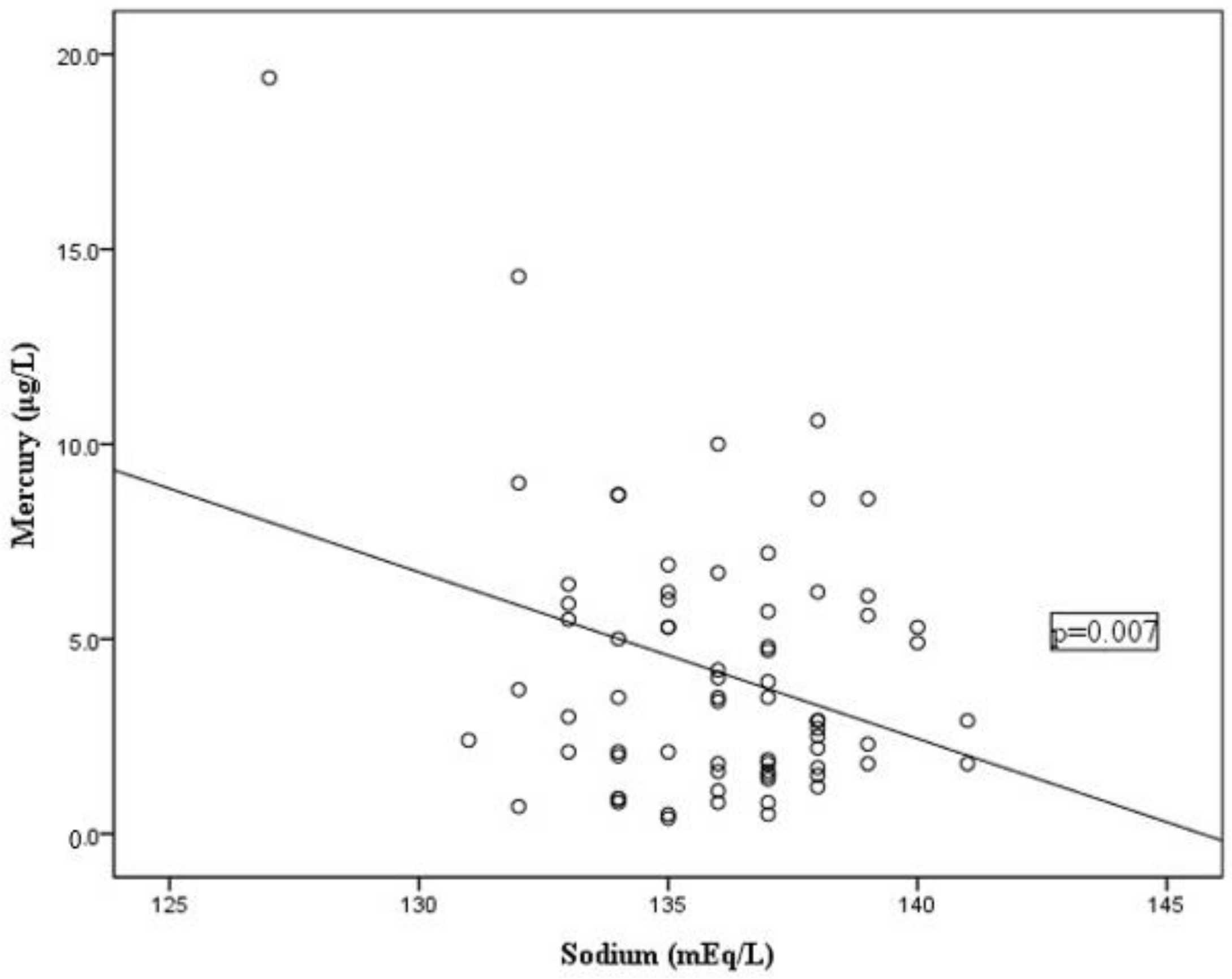
| Sodium | −0.318 | 0.007 * |
| Leukocytes | 0.016 | 0.892 |
| Percentage of neutrophil | 0.076 | 0.506 |
| Percentage of lymphocyte | −0.109 | 0.340 |
| Percentage of monocyte | −0.053 | 0.642 |
| Percentage of eosinophil | −0.024 | 0.836 |
| Percentage of basophil | −0.011 | 0.927 |
| Platelet count | −0.041 | 0.721 |
| Aspartate transaminase | 0.095 | 0.406 |
| Alanine transaminase | 0.057 | 0.618 |
| C reactive protein | 0.061 | 0.596 |
| Values for Blood Mercury in School-Aged Children | ||||
|---|---|---|---|---|
| μg/L | μg/L | p-Value | ||
| Taiwan | 5.33 | Japan | 4.55 | 0.0025 |
| Japan | 4.55 | Korea | 2.12 | <0.01 |
| Korea | 2.12 | America | 0.41 | <0.01 |
| Values for Blood Mercury in Preschool-Aged Children | ||||
| Taiwanese with Kawasaki disease | 4.15 | Korea | 2.05 | <0.01 |
| Korea | 2.05 | China | 1.10 | <0.01 |
| China | 1.10 | America | 0.45 | <0.01 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, L.-S.; Yan, J.-H.; Li, J.-Y.; Yeter, D.D.; Huang, Y.-H.; Guo, M.M.-H.; Lo, M.-H.; Kuo, H.-C. Blood Mercury Levels in Children with Kawasaki Disease and Disease Outcome. Int. J. Environ. Res. Public Health 2020, 17, 3726. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17103726
Chang L-S, Yan J-H, Li J-Y, Yeter DD, Huang Y-H, Guo MM-H, Lo M-H, Kuo H-C. Blood Mercury Levels in Children with Kawasaki Disease and Disease Outcome. International Journal of Environmental Research and Public Health. 2020; 17(10):3726. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17103726
Chicago/Turabian StyleChang, Ling-Sai, Jia-Huei Yan, Jin-Yu Li, Deniz Des Yeter, Ying-Hsien Huang, Mindy Ming-Huey Guo, Mao-Hung Lo, and Ho-Chang Kuo. 2020. "Blood Mercury Levels in Children with Kawasaki Disease and Disease Outcome" International Journal of Environmental Research and Public Health 17, no. 10: 3726. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17103726






