Overview on Osteoporosis, Periodontitis and Oral Dysbiosis: The Emerging Role of Oral Microbiota
Abstract
:1. Introduction
2. Osteoporosis
3. Periodontitis
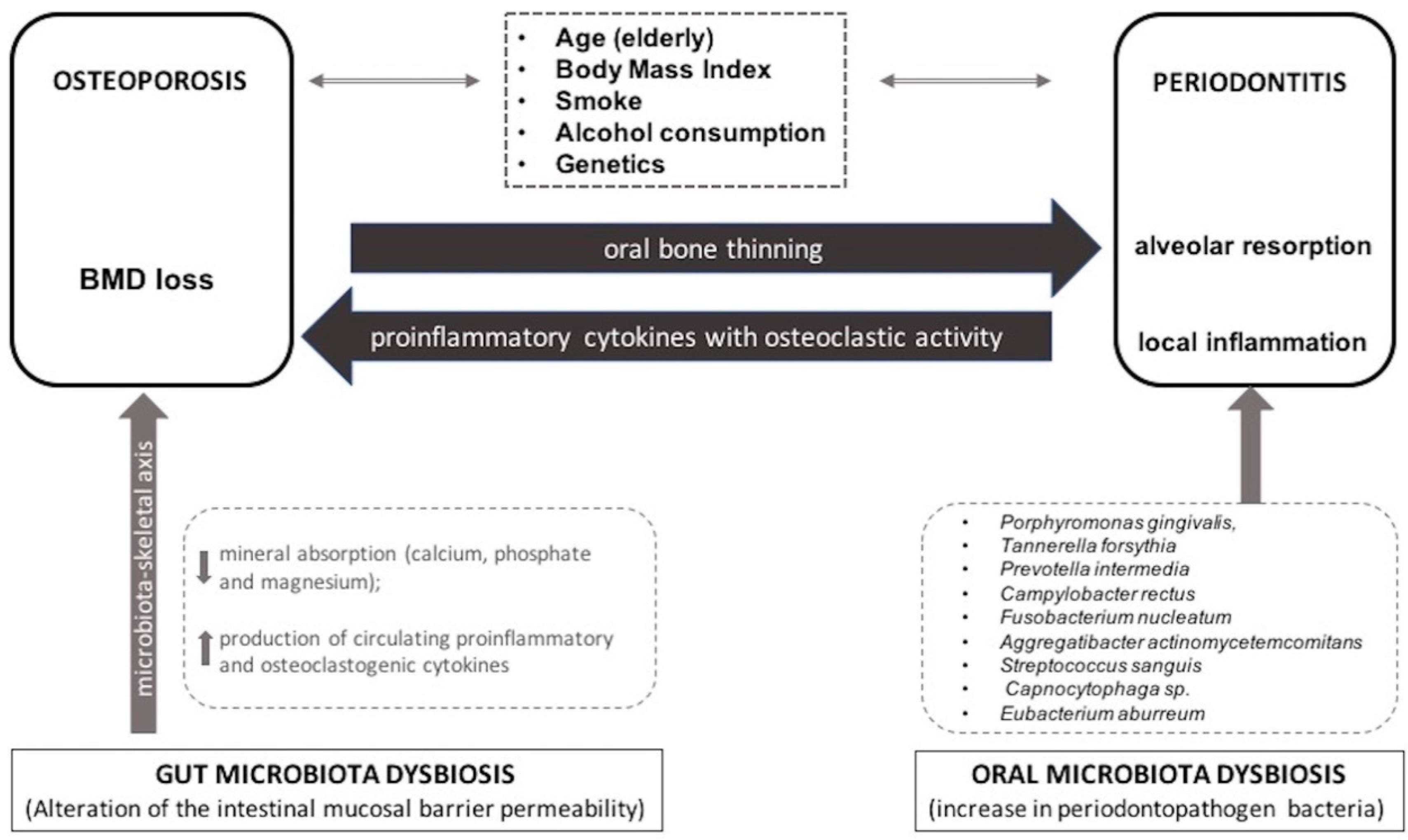
4. Osteoporosis and Periodontitis
5. Periodontitis and Osteoporosis
6. Microbiome, Dysbiosis and Osteoporosis
6.1. Gut Microbiome/Microbiota and OP
- influencing the absorption of mineral (calcium, phosphate and magnesium) [60];
6.2. Oral Microbiome/Microbiota and OP
7. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Della Vella, F.; Contaldo, M.; Fucile, R.; Panza, F.; Dibello, V.; Kalemaj, Z.; Ninivaggi, R.; Petruzzi, M.; Serpico, R. ORO-Dental Manifestations in West Syndrome. Curr. Top. Med. Chem. 2019, 19, 2824–2828. [Google Scholar] [CrossRef] [PubMed]
- Barillari, M.R.; Karali, M.; Di Iorio, V.; Contaldo, M.; Piccolo, V.; Esposito, M.; Costa, G.; Argenziano, G.; Serpico, R.; Carotenuto, M.; et al. Mild form of Zellweger Spectrum Disorders (ZSD) due to variants in PEX1: Detailed clinical investigation in a 9-years-old female. Mol. Genet. Metab. Rep. 2020, 24, 100615. [Google Scholar] [CrossRef] [PubMed]
- Contaldo, M.; Luzzi, V.; Ierardo, G.; Raimondo, E.; Boccellino, M.; Ferati, K.; Bexheti-Ferati, A.; Inchingolo, F.; Di Domenico, M.; Serpico, R.; et al. Bisphosphonate-related osteonecrosis of the jaws and dental surgery procedures in children and young people with osteogenesis imperfecta: A systematic review. J. Stomatol. Oral Maxillofac. Surg. 2020, 2468, Advance online publication. [Google Scholar] [CrossRef] [PubMed]
- De Benedittis, M.; Petruzzi, M.; Favia, G.; Serpico, R. Oro-dental manifestations in Hallopeau-Siemens-type recessive dystrophic epidermolysis bullosa. Clin. Exp. Dermatol. 2004, 29, 128–132. [Google Scholar] [CrossRef]
- Pastore, L.; Carroccio, A.; Compilato, D.; Panzarella, V.; Serpico, R.; Lo Muzio, L. Oral manifestations of celiac disease. J. Clin. Gastroenterol. 2008, 42, 224–232. [Google Scholar] [CrossRef]
- Pastore, L.; De Benedittis, M.; Petruzzi, M.; Tatò, D.; Napoli, C.; Montagna, M.T.; Catassi, C.; Serpico, R. Importanza dei segni orali nella diagnosi di forme atipiche di malattia celiaca [Importance of oral signs in the diagnosis of atypical forms of celiac disease]. Recenti Progress. Med. 2004, 95, 482–490. [Google Scholar]
- Rea, F.; Serpico, R.; Pluvio, R.; Busciolano, M.; Iovene, A.; Femiano, F.; Sessa, G.; Belnome, G. Ipoplasia dello smalto dentario in un gruppo di soggetti celiaci. Correlazioni clinico-epidemiologiche [Dental enamel hypoplasia in a group of celiac disease patients. Clinico-epidemiologic correlations]. Minerva Stomatol. 1997, 46, 517–524. [Google Scholar]
- Contaldo, M.; Romano, A.; Mascitti, M.; Fiori, F.; Della Vella, F.; Serpico, R.; Santarelli, A. Association between denture stomatitis, candida species and diabetic status. J. Biol. Regul. Homeost. Agents 2019, 33, 35–41. [Google Scholar]
- Paoletti, I.; Fusco, A.; Grimaldi, E.; Perillo, L.; Coretti, L.; Di Domenico, M.; Cozza, V.; Contaldo, M.; Serpico, R.; Guida, A.; et al. Assessment of host defence mechanisms induced by Candida species. Int. J. Immunopathol. Pharmacol. 2013, 26, 663–672. [Google Scholar] [CrossRef] [Green Version]
- Togni, L.; Mascitti, M.; Santarelli, A.; Contaldo, M.; Romano, A.; Serpico, R.; Rubini, C. Unusual Conditions Impairing Saliva Secretion: Developmental Anomalies of Salivary Glands. Front. Physiol. 2019, 10, 855. [Google Scholar] [CrossRef] [Green Version]
- Carinci, F.; Martinelli, M.; Contaldo, M.; Santoro, R.; Pezzetti, F.; Lauritano, D.; Candotto, V.; Mucchi, D.; Palmieri, A.; Tagliabue, A.; et al. Focus on periodontal disease and development of endocarditis. J. Biol. Regul. Homeost. Agents 2018, 32, 143–147. [Google Scholar] [PubMed]
- Della Vella, F.; Lauritano, D.; Lajolo, C.; Lucchese, A.; Di Stasio, D.; Contaldo, M.; Serpico, R.; Petruzzi, M. The pseudolesions of the oral mucosa: Differential diagnosis and related systemic conditions. Appl. Sci. 2019, 9, 2412. [Google Scholar] [CrossRef] [Green Version]
- Xu, Z.; Xie, Z.; Sun, J.; Huang, S.; Chen, Y.; Li, C.; Sun, X.; Xia, B.; Tian, L.; Guo, C.; et al. Gut Microbiome Reveals Specific Dysbiosis in Primary Osteoporosis. Front. Cell Infect. Microbiol. 2020, 10, 160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- NIH Consensus Development Panel on Osteoporosis Prevention, Diagnosis, and Therapy. Osteoporosis prevention, diagnosis, and therapy. JAMA 2001, 285, 785–795. [Google Scholar] [CrossRef] [PubMed]
- Contaldo, M.; Boccellino, M.; Zannini, G.; Romano, A.; Sciarra, A.; Sacco, A.; Settembre, G.; Coppola, M.; Di Carlo, A.; D’Angelo, L.; et al. Sex Hormones and Inflammation Role in Oral Cancer Progression: A Molecular and Biological Point of View. J. Oncol. 2020, 9587971. [Google Scholar] [CrossRef]
- Di Domenico, M.; Pinto, F.; Quagliuolo, L.; Contaldo, M.; Settembre, G.; Romano, A.; Coppola, M.; Ferati, K.; Bexheti-Ferati, A.; Sciarra, A.; et al. The Role of Oxidative Stress and Hormones in Controlling Obesity. Front. Endocrinol. (Lausanne) 2019, 10, 540. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sardaro, N.; Della Vella, F.; Incalza, M.A.; DI Stasio, D.; Lucchese, A.; Contaldo, M.; Laudadio, C.; Petruzzi, M. Oxidative Stress and Oral Mucosal Diseases: An Overview. In Vivo 2019, 33, 289–296. [Google Scholar] [CrossRef] [Green Version]
- Penoni, D.C.; Vettore, M.V.; Torres, S.R.; Farias, M.; Leão, A. An investigation of the bidirectional link between osteoporosis and periodontitis. Arch. Osteoporos. 2019, 14, 94. [Google Scholar] [CrossRef]
- Adler, R.A. Update on osteoporosis in men. Best Pr. Res. Clin. Endocrinol. Metab. 2018, 32, 759–772. [Google Scholar] [CrossRef]
- Colón-Emeric, C.S.; Pieper, C.F.; Van Houtven, C.H.; Grubber, J.M.; Lyles, K.W.; Lafleur, J.; Adler, R.A. Limited Osteoporosis Screening Effectiveness Due to Low Treatment Rates in a National Sample of Older Men. Mayo Clin. Proc. 2018, 93, 1749–1759. [Google Scholar] [CrossRef]
- Kanis, J.A. Assessment of fracture risk and its application to screening for postmenopausal osteoporosis: Synopsis of a WHO report. WHO Study Group. Osteoporos. Int. 1994, 4, 368–381. [Google Scholar] [CrossRef]
- Sözen, T.; Özışık, L.; Başaran, N.Ç. An overview and management of osteoporosis. Eur. J. Rheumatol. 2017, 4, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Das, M.; Cronin, O.; Keohane, D.M.; Cormac, E.M.; Nugent, H.; Nugent, M.; Molloy, C.; O’Toole, P.W.; Shanahan, F.; Molloy, M.G.; et al. Gut microbiota alterations associated with reduced bone mineral density in older adults. Rheumatol. (Oxf. Engl.) 2019, 58, 2295–2304. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wright, N.C.; Looker, A.C.; Saag, K.G.; Curtis, J.R.; Delzell, E.S.; Randall, S.; Dawson-Hughes, B. The recent prevalence of osteoporosis and low bone mass in the United States based on bone mineral density at the femoral neck or lumbar spine. J. Bone Miner. Res. 2014, 29, 2520–2526. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hernlund, E.; Svedbom, A.; Ivergård, M.; Compston, J.; Cooper, C.; Stenmark, J.; McCloskey, E.V.; Jönsson, B.; Kanis, J.A. Osteoporosis in the European Union: Medical management, epidemiology and economic burden: A report prepared in collaboration with the International Osteoporosis Foundation (IOF) and the European Federation of Pharmaceutical Industry Associations (EFPIA). Arch. Osteoporos. 2013, 8, 136. [Google Scholar] [CrossRef] [Green Version]
- Thambiah, S.C.; Yeap, S.S. Osteoporosis in South-East Asian Countries. Clin. Biochem. Rev. 2020, 41, 29–40. [Google Scholar] [CrossRef]
- Hernández-Vigueras, S.; Martínez-Garriga, B.; Sánchez, M.C.; Sanz, M.; Estrugo-Devesa, A.; Vinuesa, T.; López-López, J.; Viñas, M. Oral Microbiota, Periodontal Status, and Osteoporosis in Postmenopausal Females. J. Periodontol. 2016, 87, 124–133. [Google Scholar] [CrossRef] [Green Version]
- Slot, D.E.; Valkenburg, C.; Van der Weijden, G. Mechanical plaque removal of periodontal maintenance patients: A systematic review and network meta-analysis. J. Clin. Periodontol. 2020, 47, 107–124. [Google Scholar] [CrossRef]
- Wang, C.J.; McCauley, L.K. Osteoporosis and Periodontitis. Curr. Osteoporos. Rep. 2016, 14, 284–291. [Google Scholar] [CrossRef] [Green Version]
- Mombelli, A. Microbial colonization of the periodontal pocket and its significance for periodontal therapy. J. Periodontol. 2000, 76, 85–96. [Google Scholar] [CrossRef]
- Hienz, S.A.; Paliwal, S.; Ivanovski, S. Mechanisms of Bone Resorption in Periodontitis. J. Immunol. Res. 2015, 615486. [Google Scholar] [CrossRef] [Green Version]
- Loesche, W.J. Microbiology of Dental Decay and Periodontal Disease. In Medical Microbiology, 4th ed.; Baron, S., Ed.; University of Texas Medical Branch: Galveston Galveston, TX, USA, 1996; Chapter 99. [Google Scholar]
- Ballini, A.; Cantore, S.; Farronato, D.; Cirulli, N.; Inchingolo, F.; Papa, F.; Malcangi, G.; Inchingolo, A.D.; Dipalma, G.; Sardaro, N.; et al. Periodontal disease and bone pathogenesis: The crosstalk between cytokines and porphyromonas gingivalis. J. Biol. Regul. Homeost. Agents 2015, 29, 273–281. [Google Scholar] [PubMed]
- Gera, I. Az osteoporosis mint a destruktív fogágybetegség egyik rizikótényezóje (irodalmi áttekintés) [Osteoporosis: A risk factor for periodontal disease (literature review)]. Fogorv. Szle. 2002, 95, 49–54. [Google Scholar]
- Lauritano, D.; Moreo, G.; Carinci, F.; Borgia, R.; Lucchese, A.; Contaldo, M.; Della Vella, F.; Bernardelli, P.; Moreo, G.; Petruzzi, M. Aging and Oral Care: An Observational Study of Characteristics and Prevalence of Oral Diseases in an Italian Cohort. Int. J. Environ. Res. Public Health 2019, 16, 3763. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pepelassi, E.; Nicopoulou-Karayianni, K.; Archontopoulou, A.D.; Mitsea, A.; Kavadella, A.; Tsiklakis, K.; Vrotsos, I.; Devlin, H.; Horner, K. The relationship between osteoporosis and periodontitis in women aged 45-70 years. Oral Dis. 2012, 18, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Zhang, G.; Guo, J.F.; Tan, Y.H. Associations between Osteoporosis and risk of Periodontitis: A Pooled Analysis of Observational Studies. Oral Dis. 2020. Advance online publication. [Google Scholar] [CrossRef]
- Manjunath, S.H.; Rakhewar, P.; Nahar, P.; Tambe, V.; Gabhane, M.; Kharde, A. Evaluation of the Prevalence and Severity of Periodontal Diseases between Osteoporotic and Nonosteoporotic Subjects: A Cross-sectional Comparative Study. J. Contemp. Dent. Pr. 2019, 20, 1223–1228. [Google Scholar] [CrossRef]
- Barbato, L.; Francioni, E.; Bianchi, M.; Mascitelli, E.; Marco, L.B.; Tonelli, D.P. Periodontitis and bone metabolism. Clin. Cases Miner. Bone Metab. 2015, 12, 174–177. [Google Scholar] [CrossRef]
- Lerner, U.H. Inflammation-induced bone remodeling in periodontal disease and the influence of post-menopausal osteoporosis. J. Dent. Res. 2006, 85, 596–607. [Google Scholar] [CrossRef]
- Cantore, S.; Mirgaldi, R.; Ballini, A.; Coscia, M.F.; Scacco, S.; Papa, F.; Inchingolo, F.; Dipalma, G.; De Vito, D. Cytokine gene polymorphisms associate with microbiogical agents in periodontal disease: Our experience. Int. J. Med. Sci. 2014, 11, 674–679. [Google Scholar] [CrossRef] [Green Version]
- Inchingolo, F.; Martelli, F.S.; Gargiulo Isacco, C.; Borsani, E.; Cantore, S.; Corcioli, F.; Boddi, A.; Nguyễn, K.; De Vito, D.; Aityan, S.K.; et al. Chronic Periodontitis and Immunity, Towards the Implementation of a Personalized Medicine: A Translational Research on Gene Single Nucleotide Polymorphisms (SNPs) Linked to Chronic Oral Dysbiosis in 96 Caucasian Patients. Biomedicines 2020, 8, 115. [Google Scholar] [CrossRef] [PubMed]
- Potempa, J.; Mydel, P.; Koziel, J. The case for periodontitis in the pathogenesis of rheumatoid arthritis. Nat. Rev. Rheumatol. 2017, 13, 606–620. [Google Scholar] [CrossRef] [PubMed]
- Rydén, L.; Buhlin, K.; Ekstrand, E.; de Faire, U.; Gustafsson, A.; Holmer, J.; Kjellström, B.; Lindahl, B.; Norhammar, A.; Nygren, Å.; et al. Periodontitis Increases the Risk of a First Myocardial Infarction: A Report From the PAROKRANK Study. Circulation 2016, 133, 576–583. [Google Scholar] [CrossRef] [PubMed]
- Holmstrup, P.; Damgaard, C.; Olsen, I.; Klinge, B.; Flyvbjerg, A.; Nielsen, C.H.; Hansen, P.R. Comorbidity of periodontal disease: Two sides of the same coin? An introduction for the clinician. J. Oral. Microbiol. 2017, 9, 1332710. [Google Scholar] [CrossRef] [PubMed]
- Dominy, S.S.; Lynch, C.; Ermini, F.; Benedyk, M.; Marczyk, A.; Konradi, A.; Nguyen, M.; Haditsch, U.; Raha, D.; Griffin, C.; et al. Porphyromonas gingivalis in Alzheimer’s disease brains: Evidence for disease causation and treatment with small-molecule inhibitors. Sci. Adv. 2019, 5, eaau3333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singhrao, S.K.; Olsen, I. Assessing the role of Porphyromonas gingivalis in periodontitis to determine a causative relationship with Alzheimer’s disease. J. Oral Microbiol. 2019, 11, 1563405. [Google Scholar] [CrossRef] [Green Version]
- Pritchard, A.B.; Crean, S.; Olsen, I.; Singhrao, S.K. Periodontitis, Microbiomes and their Role in Alzheimer’s Disease. Front. Aging NeuroSci. 2017, 9, 336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Collins, A. Mapping in the sequencing era. Hum. Hered. 2000, 50, 76–84. [Google Scholar] [CrossRef]
- Lederberg, J.; McCray, A.T. ‘Ome Sweet’ Omics—A Genealogical Treasury of Words Genealogical Treasury of Words. Scientist 2001, 15, 8. [Google Scholar]
- Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature 2012, 486, 207–214. [Google Scholar] [CrossRef] [Green Version]
- Nature.com: Microbiome. Available online: https://0-www-nature-com.brum.beds.ac.uk/subjects/microbiome. (accessed on 28 July 2020).
- Berg, G.; Rybakova, D.; Fischer, D.; Cernava, T.; Vergès, M.C.; Charles, T.; Chen, X.; Cocolin, L.; Eversole, K.; Corral, G.H.; et al. Microbiome definition re-visited: Old concepts and new challenges. Microbiome 2020, 8, 103. [Google Scholar] [CrossRef] [PubMed]
- Baquero, F.; Nombela, C. The microbiome as a human organ. Clin. Microbiol. Infect. 2012, 18, 2–4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dewhirst, F.E.; Chen, T.; Izard, J.; Paster, B.J.; Tanner, A.C.; Yu, W.H.; Lakshmanan, A.; Wade, W.G. The human oral microbiome. J. Bacteriol. 2010, 192, 5002–5017. [Google Scholar] [CrossRef] [Green Version]
- Segata, N.; Haake, S.K.; Mannon, P.; Lemon, K.P.; Waldron, L.; Gevers, D.; Huttenhower, C.; Izard, J. Composition of the adult digestive tract bacterial microbiome based on seven mouth surfaces, tonsils, throat and stool samples. Genome Biol. 2012, 13, R42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wade, W.G.; Prosdocimi, E.M. Profiling of Oral Bacterial Communities. J. Dent. Res. 2020, 99, 621–629. [Google Scholar] [CrossRef]
- Behera, J.; Ison, J.; Tyagi, S.C.; Tyagi, N. The role of gut microbiota in bone homeostasis. Bone 2020, 135, 115317. [Google Scholar] [CrossRef]
- Chen, Y.C.; Greenbaum, J.; Shen, H.; Deng, H.W. Association Between Gut Microbiota and Bone Health: Potential Mechanisms and Prospective. J. Clin. Endocrinol. Metab. 2017, 102, 3635–3646. [Google Scholar] [CrossRef]
- Gargiulo Isacco, C.; Ballini, A.; Paduanelli, G.; Inchingolo, A.D.; Nguyen, K.; Inchingolo, A.M.; Pham, V.H.; Aityan, S.K.; Schiffman, M.; Tran, T.C.; et al. Bone decay and beyond: How can we approach it better. J. Biol. Regul. Homeost. Agents 2019, 33, 143–154. [Google Scholar]
- Liang, W.; Li, X.; Li, Y.; Li, C.; Gao, B.; Gan, H.; Li, S.; Shen, J.; Kang, J.; Ding, S.; et al. Tongue coating microbiome regulates the changes in tongue texture and coating in patients with post-menopausal osteoporosis of Gan-shen deficiency syndrome type. Int. J. Mol. Med. 2013, 32, 1069–1076. [Google Scholar] [CrossRef] [Green Version]
- Banack, H.R.; Genco, R.J.; LaMonte, M.J.; Millen, A.E.; Buck, M.J.; Sun, Y.; Andrews, C.A.; Hovey, K.M.; Tsompana, M.; McSkimming, D.I.; et al. Cohort profile: The Buffalo OsteoPerio microbiome prospective cohort study. BMJ Open 2018, 8, e024263. [Google Scholar] [CrossRef]
- Brennan, R.M.; Genco, R.J.; Wilding, G.E.; Hovey, K.M.; Trevisan, M.; Wactawski-Wende, J. Bacterial species in subgingival plaque and oral bone loss in postmenopausal women. J. Periodontol. 2007, 78, 1051–1061. [Google Scholar] [CrossRef] [PubMed]
- Rizzoli, R. Nutritional influence on bone: Role of gut microbiota. Aging Clin. Exp. Res. 2019, 31, 743–751. [Google Scholar] [CrossRef]
- Morato-Martínez, M.; López-Plaza, B.; Santurino, C.; Palma-Milla, S.; Gómez-Candela, C. A Dairy Product to Reconstitute Enriched with Bioactive Nutrients Stops Bone Loss in High-Risk Menopausal Women without Pharmacological Treatment. Nutrients 2020, 12, 2203. [Google Scholar] [CrossRef] [PubMed]
- Zaura, E.; Twetman, S. Critical Appraisal of Oral Pre- and Probiotics for Caries Prevention and Care. Caries Res. 2019, 53, 514–526. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.; Brody, H.; Lin, G.H.; Rangé, H.; Kuraji, R.; Ye, C.; Kamarajan, P.; Radaic, A.; Gao, L.; Kapila, Y. Probiotics, including nisin-based probiotics, improve clinical and microbial outcomes relevant to oral and systemic diseases. J. Periodontol. 2000 2020, 82, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Huck, O.; Mulhall, H.; Rubin, G.; Kizelnik, Z.; Iyer, R.; Perpich, J.D.; Haque, N.; Cani, P.D.; de Vos, W.M.; Amar, S. Akkermansia muciniphila reduces Porphyromonas gingivalis-induced inflammation and periodontal bone destruction. J. Clin. Periodontol. 2020, 47, 202–212. [Google Scholar] [CrossRef]
- Slomka, V.; Hernandez-Sanabria, E.; Herrero, E.R.; Zaidel, L.; Bernaerts, K.; Boon, N.; Quirynen, M.; Teughels, W. Nutritional stimulation of commensal oral bacteria suppresses pathogens: The prebiotic concept. J. Clin. Periodontol. 2017, 44, 344–352. [Google Scholar] [CrossRef]
- Hirasawa, M.; Kurita-Ochia, T. Probiotic Potential of Lactobacilli Isolated from Saliva of Periodontally Healthy Individuals. Oral Health Prev. Dent. 2020, 18, 563–570. [Google Scholar] [CrossRef]
- Rosier, B.T.; Buetas, E.; Moya-Gonzalvez, E.M.; Artacho, A.; Mira, A. Nitrate as a potential prebiotic for the oral microbiome. Sci. Rep. 2020, 10, 12895. [Google Scholar] [CrossRef]
- Pudgar, P.; Povšič, K.; Čuk, K.; Seme, K.; Petelin, M.; Gašperšič, R. Probiotic strains of Lactobacillus brevis and Lactobacillus plantarum as adjunct to non-surgical periodontal therapy: 3-month results of a randomized controlled clinical trial. Clin. Oral Investig. 2020. Advance online publication. [Google Scholar] [CrossRef]
- Cantore, S.; Ballini, A.; De Vito, D.; Abbinante, A.; Altini, V.; Dipalma, G.; Inchingolo, F.; Saini, R. Clinical results of improvement in periodontal condition by administration of oral probiotics. J. Biol. Regul. Homeost. Agents 2018, 32, 1329–1334. [Google Scholar]
- Inchingolo, F.; Dipalma, G.; Cirulli, N.; Cantore, S.; Saini, R.S.; Altini, V.; Santacroce, L.; Ballini, A.; Saini, R. Microbiological results of improvement in periodontal condition by administration of oral probiotics. J. Biol. Regul. Homeost. Agents 2018, 32, 1323–1328. [Google Scholar] [PubMed]
- Matsubara, V.H.; Bandara, H.M.; Ishikawa, K.H.; Mayer, M.P.; Samaranayake, L.P. The role of probiotic bacteria in managing periodontal disease: A systematic review. Expert Rev. Anti-Infect. Ther. 2016, 14, 643–655. [Google Scholar] [CrossRef] [PubMed]
- Bustamante, M.; Oomah, B.D.; Mosi-Roa, Y.; Rubilar, M.; Burgos-Díaz, C. Probiotics as an Adjunct Therapy for the Treatment of Halitosis, Dental Caries and Periodontitis. Probiotics Antimicrob. Proteins 2020, 12, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Hanel, A.N.; Herzog, H.M.; James, M.G.; Cuadra, G.A. Effects of Oral Commensal Streptococci on Porphyromonas gingivalis Invasion into Oral Epithelial Cells. Dent. J. (Basel) 2020, 8, 39. [Google Scholar] [CrossRef] [PubMed]
- Ohshima, J.; Wang, Q.; Fitzsimonds, Z.R.; Miller, D.P.; Sztukowska, M.N.; Jung, Y.J.; Hayashi, M.; Whiteley, M.; Lamont, R.J. Streptococcus gordonii programs epithelial cells to resist ZEB2 induction by Porphyromonas gingivalis. Proc. Natl. Acad. Sci. USA 2019, 116, 8544–8553. [Google Scholar] [CrossRef] [Green Version]
- Ho, M.H.; Lamont, R.J.; Xie, H. Identification of Streptococcus cristatus peptides that repress expression of virulence genes in Porphyromonas gingivalis. Sci. Rep. 2017, 7, 1413. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Contaldo, M.; Itro, A.; Lajolo, C.; Gioco, G.; Inchingolo, F.; Serpico, R. Overview on Osteoporosis, Periodontitis and Oral Dysbiosis: The Emerging Role of Oral Microbiota. Appl. Sci. 2020, 10, 6000. https://0-doi-org.brum.beds.ac.uk/10.3390/app10176000
Contaldo M, Itro A, Lajolo C, Gioco G, Inchingolo F, Serpico R. Overview on Osteoporosis, Periodontitis and Oral Dysbiosis: The Emerging Role of Oral Microbiota. Applied Sciences. 2020; 10(17):6000. https://0-doi-org.brum.beds.ac.uk/10.3390/app10176000
Chicago/Turabian StyleContaldo, Maria, Annalisa Itro, Carlo Lajolo, Gioele Gioco, Francesco Inchingolo, and Rosario Serpico. 2020. "Overview on Osteoporosis, Periodontitis and Oral Dysbiosis: The Emerging Role of Oral Microbiota" Applied Sciences 10, no. 17: 6000. https://0-doi-org.brum.beds.ac.uk/10.3390/app10176000





