Urolithin A and B Alter Cellular Metabolism and Induce Metabolites Associated with Apoptosis in Leukemic Cells
Abstract
:1. Introduction
2. Results
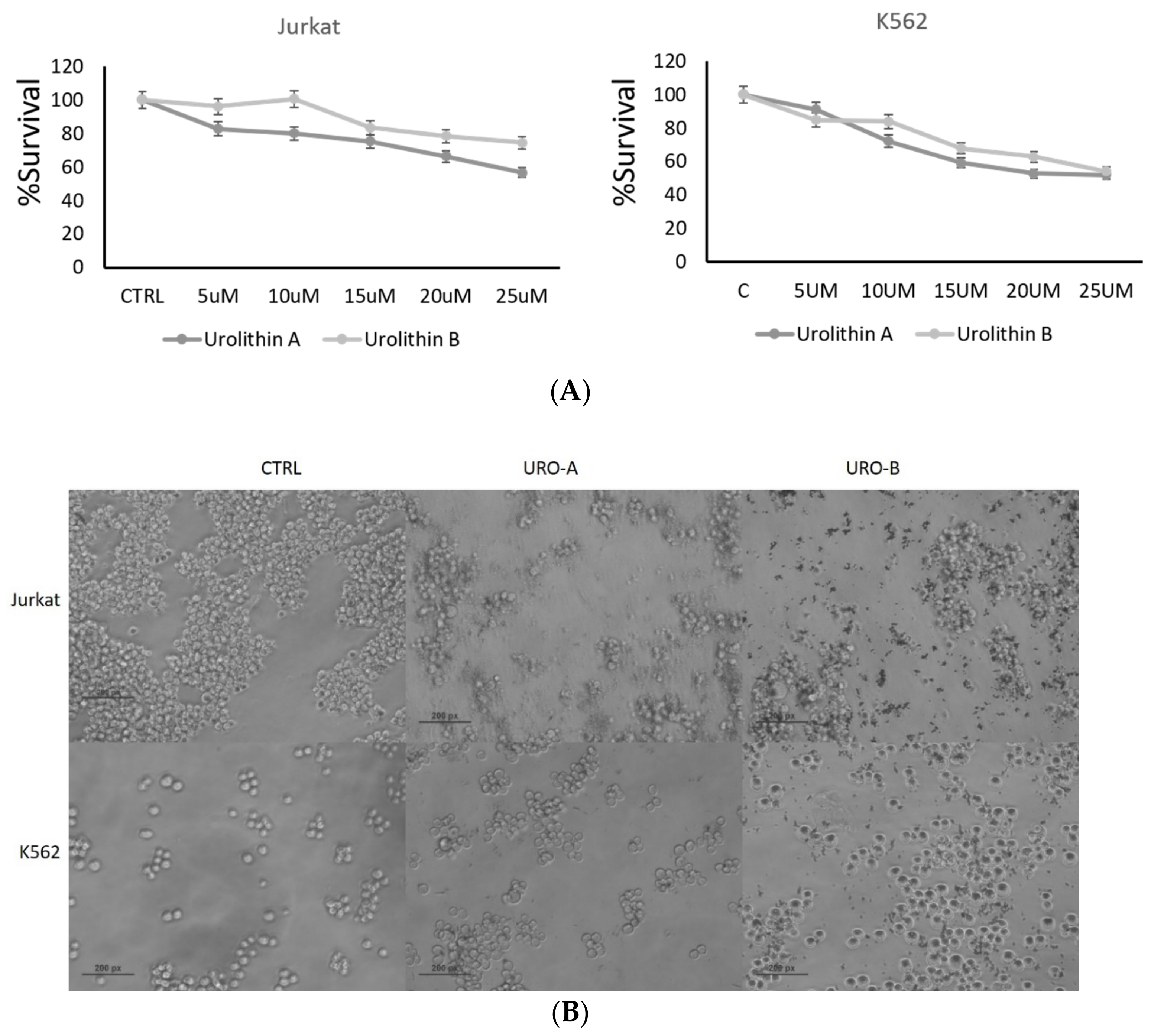
2.1. Urolithin A and B Treatments Inhibit Leukemic Cell Proliferation
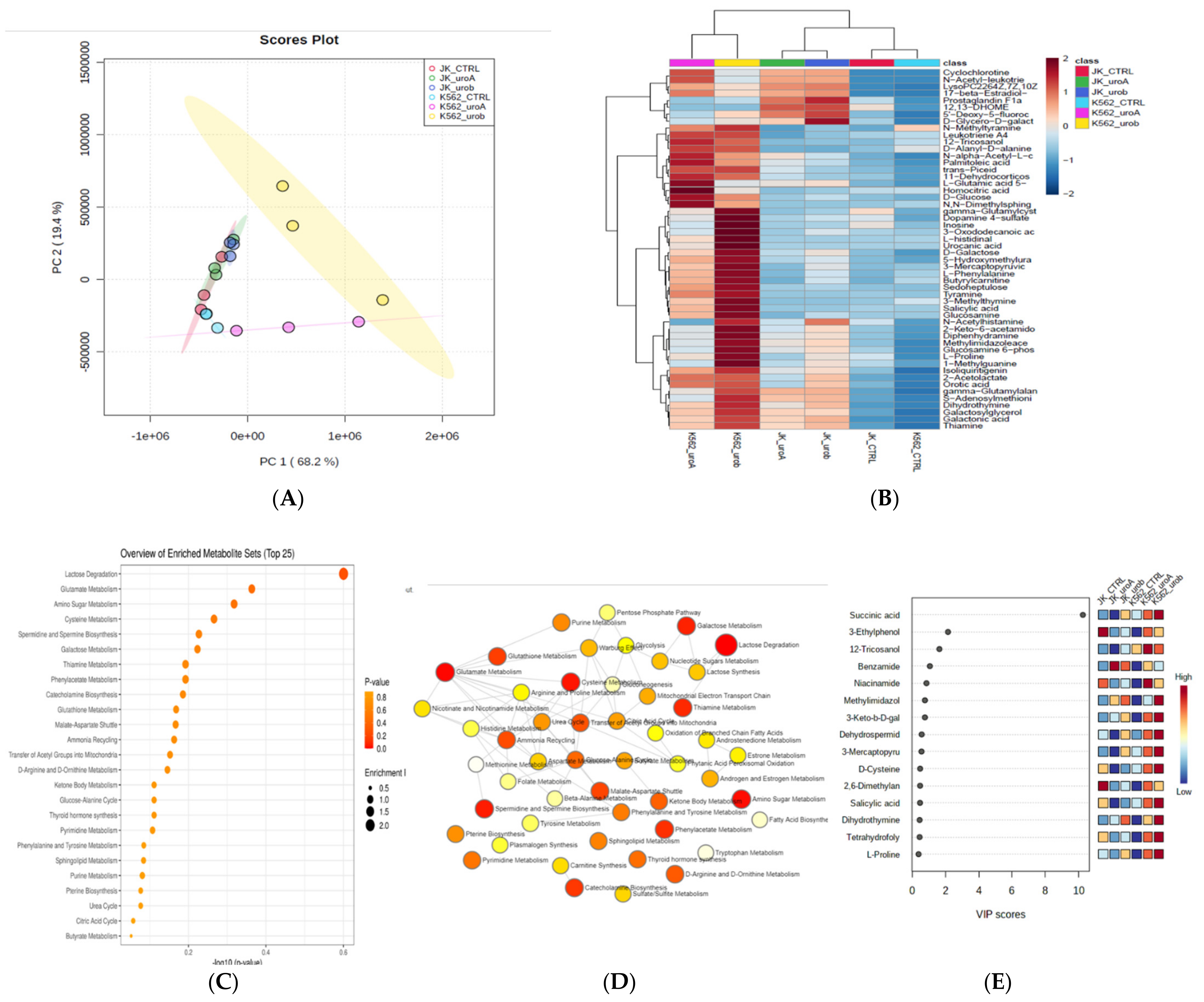
2.2. Urolithin A and B Alter the Global Metabolic Landscape of Leukemic Cells
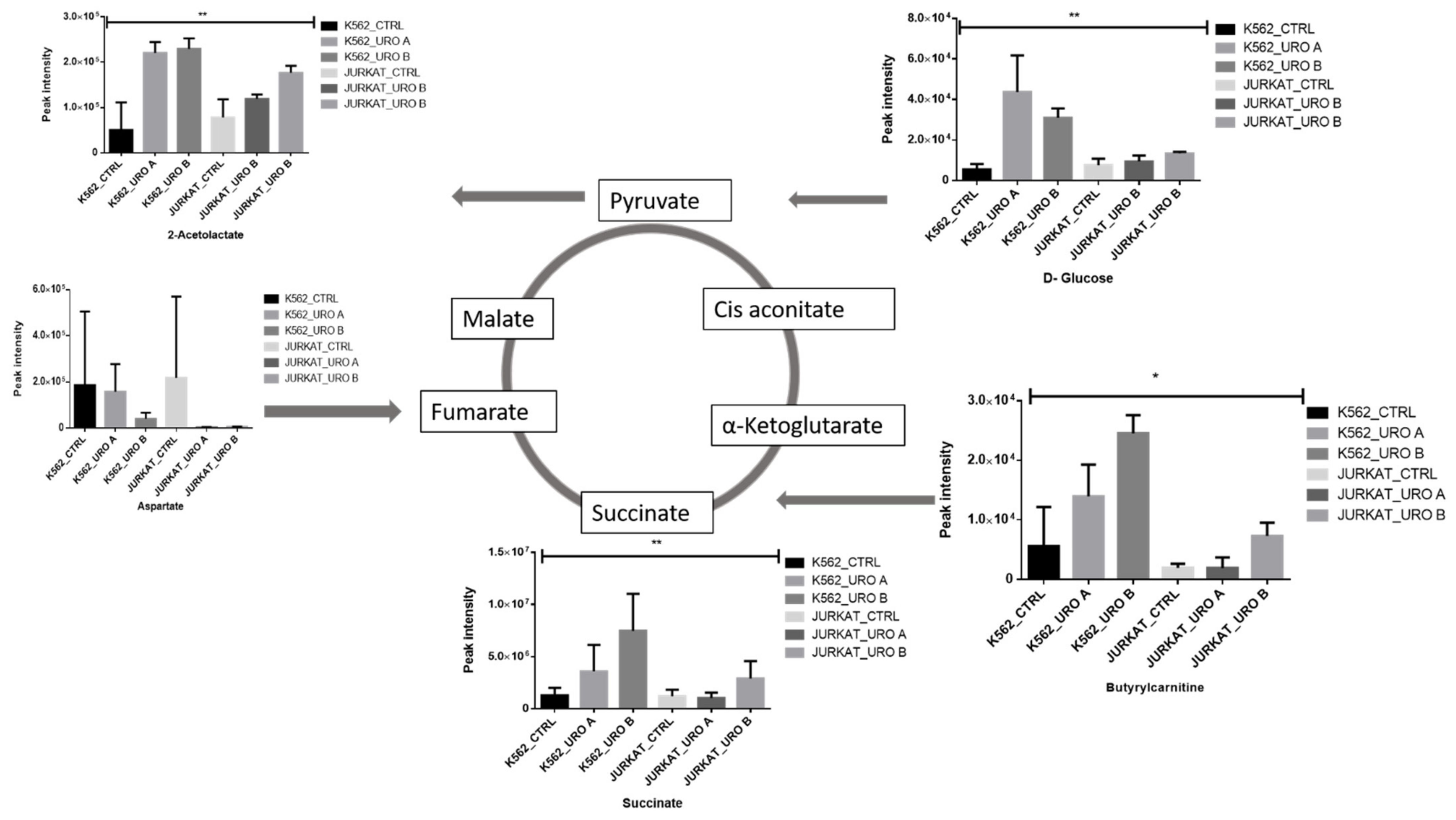
2.3. Urolithin A and B Alter Levels of Cellular Energy Metabolites
2.4. Urolithin A and B Treatments Modify Glutamine Metabolism
2.5. Urolithin A and B Treatments Modulate One-Carbon Metabolism in the Leukemic Cell Lines
2.6. Urolithin A and B Treatments Induce a Level of Fatty Acid Responsible for Cell Death
2.7. Urolithin A and B Treatments Increase the Level of DNA Damage-Associated Metabolites in Leukemic Cell Lines
3. Discussion
4. Materials and Methods
4.1. Cell Cultures and Medium Composition
4.2. Measurement of Cell Proliferation by MTT Assay
4.3. Metabolites Extraction
4.4. Mass Spectrometry
4.5. Data Analysis
4.6. Apoptosis Assay
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dahlawi, H.; Jordan-Mahy, N.; Clench, M.; McDougall, G.J.; Maitre, C.L. Polyphenols are responsible for the proapoptotic properties of pomegranate juice on leukemia cell lines. Food Sci. Nutr. 2013, 1, 196–208. [Google Scholar] [CrossRef]
- Fimognari, C.; Berti, F.; Nüsse, M.; Cantelli-Forti, G.; Hrelia, P. Induction of apoptosis in two human leukemia cell lines as well as differentiation in human promyelocytic cells by cyanidin-3-O-β-glucopyranoside. Biochem. Pharmacol. 2004, 67, 2047–2056. [Google Scholar] [CrossRef] [PubMed]
- Richardson, P.G.; on behalf of the International Myeloma Working Group; Delforge, M.; Beksac, M.; Wen, P.; Jongen, J.L.; Sezer, O.; Terpos, E.; Munshi, N.; Palumbo, A.; et al. Management of treatment-emergent peripheral neuropathy in multiple myeloma. Leukemia 2011, 26, 595–608. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suttorp, M.; Eckardt, L.; Tauer, J.T.; Millot, F. Management of Chronic Myeloid Leukemia in Childhood. Curr. Hematol. Malign Rep. 2012, 7, 116–124. [Google Scholar] [CrossRef]
- Shanshal, M.; Haddad, R.Y. Chronic Lymphocytic Leukemia. Dis. Mon. 2012, 58, 153–167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suo, S.-S.; Li, C.-Y.; Zhang, Y.; Wang, J.-H.; Lou, Y.-J.; Yu, W.-J.; Jin, J. Characteristics of chemotherapy-induced diabetes mellitus in acute lymphoblastic leukemia patients. J. Zhejiang Univ. Sci. B 2020, 21, 740–744. [Google Scholar] [CrossRef]
- Arteaga, M.F.; Mikesch, J.-H.; Fung, T.-K.; So, C.W. Epigenetics in acute promyelocytic leukaemia pathogenesis and treatment response: A TRAnsition to targeted therapies. Br. J. Cancer 2015, 112, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Zeisig, B.B.; Kulasekararaj, A.G.; Mufti, G.J.; So, C.W.E. SnapShot: Acute Myeloid Leukemia. Cancer Cell 2012, 22, 698–698.e1. [Google Scholar] [CrossRef] [Green Version]
- Khan, N.; Afaq, F.; Mukhtar, H. Cancer Chemoprevention through Dietary Antioxidants: Progress and Promise. Antioxid. Redox Signal. 2008, 10, 475–510. [Google Scholar] [CrossRef]
- Syed, D.N.; Suh, Y.; Afaq, F.; Mukhtar, H. Dietary agents for chemoprevention of prostate cancer. Cancer Lett. 2008, 265, 167–176. [Google Scholar] [CrossRef] [Green Version]
- Paller, C.J.; Ye, X.; Wozniak, P.J.; Gillespie, B.K.; Sieber, P.R.; Greengold, R.H.; Stockton, B.R.; Hertzman, B.L.; Efros, M.D.; Roper, R.P.; et al. A randomized phase II study of pomegranate extract for men with rising PSA following initial therapy for localized prostate cancer. Prostate Cancer Prostatic Dis. 2012, 16, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Afaq, F.; Kweon, M.-H.; Kim, K.; Mukhtar, H. Oral Consumption of Pomegranate Fruit Extract Inhibits Growth and Progression of Primary Lung Tumors in Mice. Cancer Res. 2007, 67, 3475–3482. [Google Scholar] [CrossRef] [Green Version]
- Adams, L.S.; Seeram, N.P.; Aggarwal, B.B.; Takada, Y.; Sand, A.D.; Heber†, D. Pomegranate Juice, Total Pomegranate Ellagitannins, and Punicalagin Suppress Inflammatory Cell Signaling in Colon Cancer Cells. J. Agric. Food Chem. 2006, 54, 980–985. [Google Scholar] [CrossRef]
- Afaq, F.; Abu Zaid, M.; Khan, N.; Dreher, M.; Mukhtar, H. Protective effect of pomegranate-derived products on UVB-mediated damage in human reconstituted skin. Exp. Dermatol. 2009, 18, 553–561. [Google Scholar] [CrossRef] [Green Version]
- Kim, N.D.; Mehta, R.; Yu, W.; Neeman, I.; Livney, T.; Amichay, A.; Poirier, D.; Nicholls, P.; Kirby, A.; Jiang, W.; et al. Chemopreventive and adjuvant therapeutic potential of pomegranate (Punica granatum) for human breast cancer. Breast Cancer Res. Treat. 2002, 71, 203–217. [Google Scholar] [CrossRef]
- Dahlawi, H.; Jordan-Mahy, N.; Clench, M.R.; Le Maitre, C.L. Bioactive Actions of Pomegranate Fruit Extracts on Leukemia Cell Lines In Vitro Hold Promise for New Therapeutic Agents for Leukemia. Nutr. Cancer 2012, 64, 100–110. [Google Scholar] [CrossRef] [PubMed]
- Mohammed Saleem, Y.I.; Albassam, H.; Selim, M. Urolithin A induces prostate cancer cell death in p53-dependent and in p53-independent manner. Eur. J. Nutr. 2019, 59, 1607–1618. [Google Scholar] [CrossRef] [PubMed]
- Lv, M.; Shi, C.; Pan, F.; Shao, J.; Feng, L.; Chen, G.; Ou, C.; Zhang, J.; Fu, W. Urolithin B suppresses tumor growth in hepatocellular carcinoma through inducing the inactivation of Wnt/β-catenin signaling. J. Cell. Biochem. 2019, 120, 17273–17282. [Google Scholar] [CrossRef]
- Llorach, R.; Cerdá, B.; Cerón, J.J.; Espín, J.C.; Tomás-Barberán, F.A. Evaluation of the bioavailability and metabolism in the rat of punicalagin, an antioxidant polyphenol from pomegranate juice. Eur. J. Nutr. 2003, 42, 18–28. [Google Scholar] [CrossRef]
- García-Villalba, R.; Selma, M.V.; Espín, J.C.; Tomás-Barberán, F.A. Identification of Novel Urolithin Metabolites in Human Feces and Urine after the Intake of a Pomegranate Extract. J. Agric. Food Chem. 2019, 67, 11099–11107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Espín, J.C.; Larrosa, M.; García-Conesa, M.T.; Tomás-Barberán, F. Biological Significance of Urolithins, the Gut Microbial Ellagic Acid-Derived Metabolites: The Evidence So Far. Evid. Based Complement. Altern. Med. 2013, 2013, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Seeram, N.P.; Henning, S.M.; Zhang, Y.; Suchard, M.; Li, Z.; Heber, D. Pomegranate Juice Ellagitannin Metabolites Are Present in Human Plasma and Some Persist in Urine for Up to 48 Hours. J. Nutr. 2006, 136, 2481–2485. [Google Scholar] [CrossRef] [Green Version]
- De Araujo, R.L.; Tomás-Barberán, F.A.; dos Santos, R.F.; Martinez-Blazquez, J.A.; Genovese, M.I. Postprandial glucose-lowering effect of cagaita (Eugenia dysenterica DC) fruit juice in dysglycemic subjects with metabolic syndrome: An exploratory study. Food Res. Int. 2021, 142, 110209. [Google Scholar] [CrossRef]
- Timmerman, L.A.; Holton, T.; Yuneva, M.; Louie, R.J.; Padró, M.; Daemen, A.; Hu, M.; Chan, D.A.; Ethier, S.P.; Veer, L.J.V.T.; et al. Glutamine Sensitivity Analysis Identifies the xCT Antiporter as a Common Triple-Negative Breast Tumor Therapeutic Target. Cancer Cell 2013, 24, 450–465. [Google Scholar] [CrossRef] [Green Version]
- Sun, C.; Chesnokov, V.; Larson, G.; Itakura, K. Glucosamine Enhances TRAIL-Induced Apoptosis in the Prostate Cancer Cell Line DU145. Medicines 2019, 6, 104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, N.-H.; Cheong, K.A.; Kim, C.-H.; Noh, M.; Lee, A.-Y. Glucosamine Induces Activated T Cell Apoptosis through Reduced T Cell Receptor. Scand. J. Immunol. 2013, 78, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Karaskov, E.; Scott, C.; Zhang, L.; Teodoro, T.; Ravazzola, M.; Volchuk, A. Chronic Palmitate But Not Oleate Exposure Induces Endoplasmic Reticulum Stress, Which May Contribute to INS-1 Pancreatic β-Cell Apoptosis. Endocrinology 2006, 147, 3398–3407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chai, W.; Liu, Z. p38 Mitogen-Activated Protein Kinase Mediates Palmitate-Induced Apoptosis But Not Inhibitor of Nuclear Factor-κB Degradation in Human Coronary Artery Endothelial Cells. Endocrinology 2007, 148, 1622–1628. [Google Scholar] [CrossRef] [Green Version]
- Boffa, L.C.; Lupton, J.R.; Mariani, M.R.; Ceppi, M.; Newmark, H.L.; Scalmati, A.; Lipkin, M. Modulation of colonic epithelial cell proliferation, histone acetylation, and luminal short chain fatty acids by variation of dietary fiber (wheat bran) in rats. Cancer Res. 1992, 52, 5906–5912. [Google Scholar]
- Roscilli, G.; Marra, E.; Mori, F.; Di Napoli, A.; Mancini, R.; Serlupi-Crescenzi, O.; Virmani, A.; Aurisicchio, L.; Ciliberto, G. Carnitines slow down tumor development of colon cancer in the DMH-chemical carcinogenesis mouse model. J. Cell. Biochem. 2013, 114, 1665–1673. [Google Scholar] [CrossRef]
- Kotas, M.E.; Jurczak, M.J.; Annicelli, C.; Gillum, M.P.; Cline, G.W.; Shulman, G.I.; Medzhitov, R. Role of caspase-1 in regulation of triglyceride metabolism. Proc. Natl. Acad. Sci. USA 2013, 110, 4810–4815. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Byberg, L.; Kilander, L.; Lemming, E.W.; Michaelsson, K.; Vessby, B. Cancer death is related to high palmitoleic acid in serum and to polymorphisms in the SCD-1 gene in healthy Swedish men. Am. J. Clin. Nutr. 2013, 99, 551–558. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schumann, R.R.; Belka, C.; Reuter, D.; Lamping, N.; Kirschning, C.J.; Weber, J.R.; Pfeil, D. Lipopolysaccharide activates caspase-1 (interleukin-1-converting enzyme) in cultured monocytic and endothelial cells. Blood 1998, 91, 577–584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gonzalvez, F.; Schug, Z.T.; Houtkooper, R.H.; MacKenzie, E.D.; Brooks, D.G.; Wanders, R.J.A.; Petit, P.X.; Vaz, F.; Gottlieb, E. Cardiolipin provides an essential activating platform for caspase-8 on mitochondria. J. Cell Biol. 2008, 183, 681–696. [Google Scholar] [CrossRef] [Green Version]
- Němcová-Fürstová, V.; James, R.F.; Kovář, J. Inhibitory Effect of Unsaturated Fatty Acids on Saturated Fatty Acid-Induced Apoptosis in Human Pancreatic β-Cells: Activation of Caspases and ER Stress Induction. Cell. Physiol. Biochem. 2011, 27, 525–538. [Google Scholar] [CrossRef] [Green Version]
- Alghamdi, A.A.; Mohammed, M.R.S.; Zamzami, M.A.; Al-Malki, A.L.; Qari, M.H.; Khan, M.I.; Choudhry, H. Untargeted Metabolomics Identifies Key Metabolic Pathways Altered by Thymoquinone in Leukemic Cancer Cells. Nutrients 2020, 12, 1792. [Google Scholar] [CrossRef]
- Mohammed, M.R.S.; Alghamdi, R.A.; Alzahrani, A.M.; Zamzami, M.A.; Choudhry, H.; Khan, M.I. Compound C, a Broad Kinase Inhibitor Alters Metabolic Fingerprinting of Extra Cellular Matrix Detached Cancer Cells. Front. Oncol. 2021, 11. [Google Scholar] [CrossRef]
- Hassan, M.A.; Al-Sakkaf, K.; Razeeth, M.; Dallol, A.; Al-Maghrabi, J.; AlDahlawi, A.; Ashoor, S.; Maamra, M.; Ragoussis, J.; Wu, W.; et al. Integration of Transcriptome and Metabolome Provides Unique Insights to Pathways Associated With Obese Breast Cancer Patients. Front. Oncol. 2020, 10. [Google Scholar] [CrossRef]
- Nadeem, M.S.; Razeeth, M.; Choudhry, H.M.Z.; Anwar, F.; Zamzami, M.A.; Murtaza, B.N.; Al-Abbasi, F.A.M.; Khan, M.I.; Shakoori, A.R. LC-MS/MS-based metabolic profiling of Escherichia coli under heterologous gene expression stress. J. Cell. Biochem. 2019, 121, 125–134. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alzahrani, A.M.; Shait Mohammed, M.R.; Alghamdi, R.A.; Ahmad, A.; Zamzami, M.A.; Choudhry, H.; Khan, M.I. Urolithin A and B Alter Cellular Metabolism and Induce Metabolites Associated with Apoptosis in Leukemic Cells. Int. J. Mol. Sci. 2021, 22, 5465. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22115465
Alzahrani AM, Shait Mohammed MR, Alghamdi RA, Ahmad A, Zamzami MA, Choudhry H, Khan MI. Urolithin A and B Alter Cellular Metabolism and Induce Metabolites Associated with Apoptosis in Leukemic Cells. International Journal of Molecular Sciences. 2021; 22(11):5465. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22115465
Chicago/Turabian StyleAlzahrani, Abdulaziz Musa, Mohammed Razeeth Shait Mohammed, Raed Ahmed Alghamdi, Abrar Ahmad, Mazin A. Zamzami, Hani Choudhry, and Mohammad Imran Khan. 2021. "Urolithin A and B Alter Cellular Metabolism and Induce Metabolites Associated with Apoptosis in Leukemic Cells" International Journal of Molecular Sciences 22, no. 11: 5465. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22115465





