Role of Peroxisome Proliferator-Activated Receptors (PPARs) in Trophoblast Functions
Abstract
:1. Introduction
2. Biological Functions of PPARs
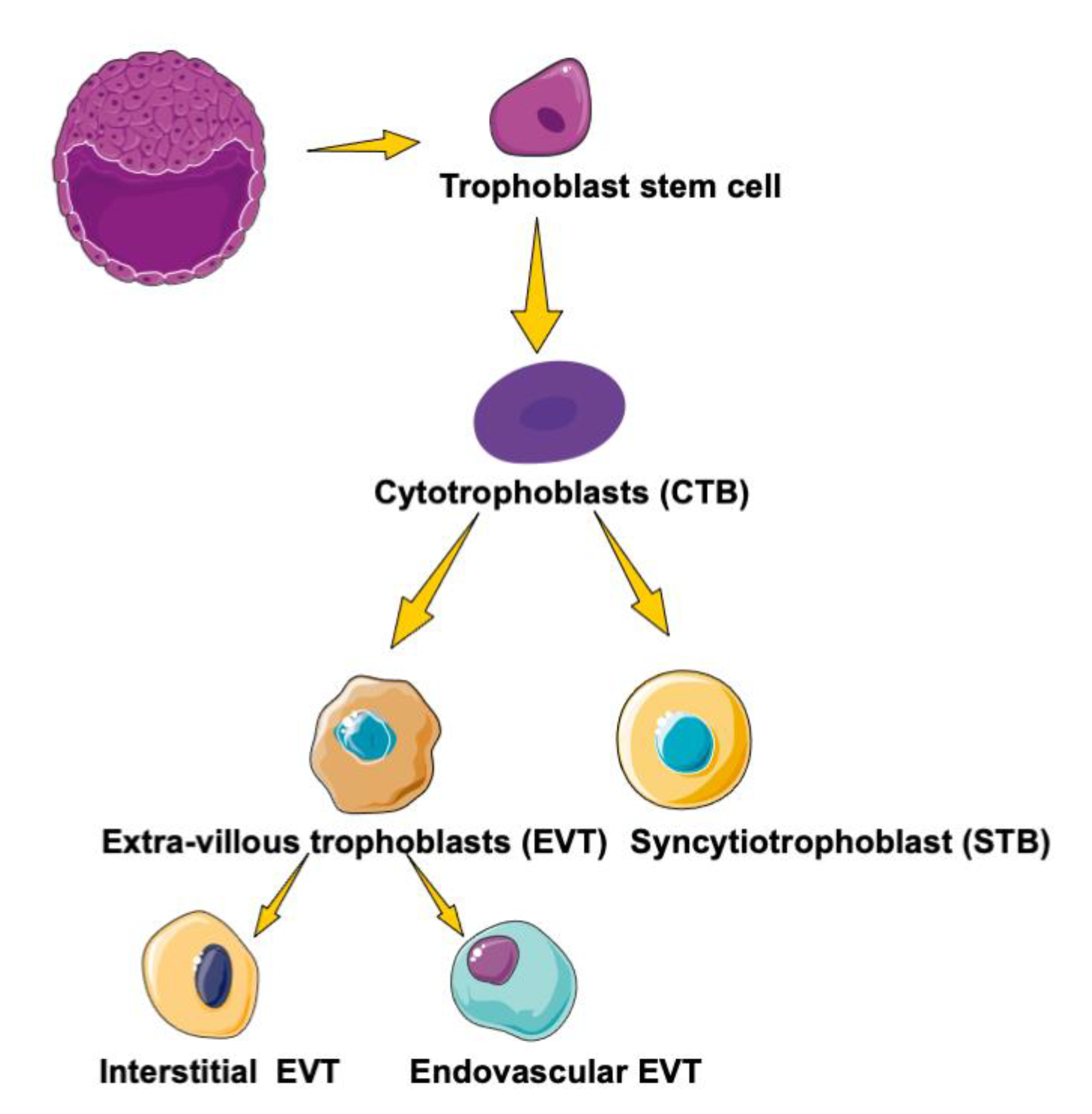
3. PPARs in Trophoblast Differentiation
4. PPARs in Other Functions of Trophoblasts
4.1. Secretion
4.2. Fusion and Maturation
4.3. Proliferation, Migration, and Invasion
5. PPARs and Energy Metabolism in the Trophoblast
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| PPARs | peroxisome proliferator-activated receptors |
| RARs | retinoic acid receptors |
| TRs | thyroid hormone receptors |
| LXRs | liver X receptors |
| VDRs | vitamin D3 receptors |
| SRs | steroid receptors |
| PPRE | PPAR responsive element |
| LC-PUFAs | long-chain polyunsaturated fatty acids |
| LTB4 | leukotriene B4 |
| PGI2 | prostaglandin I2 |
| cPGI2 | carbaprostacyclin |
| 15dPGJ2 | 15-deoxy-12,14-prostaglandin J2 |
| NLSs | nuclear localization signal |
| DBD | DNA-binding domain |
| AF1 | activation function 1 |
| ERK | extracellular signal-regulated kinase |
| MAPK | mitogen-activated protein kinase |
| TE | trophectoderm |
| CTB | cytotrophoblast |
| STB | syncytiotrophoblast |
| EVT | extravillous trophoblast |
| GCM-1 | chorion-specific transcription factor |
| hCG | chorionic gonadotropin |
| VCT | villous cytotrophoblasts |
| H-, L-FABPs | heart and liver fatty acid-binding proteins |
| HIF | hypoxia inducible factor |
| HDACs | histone deacetylases |
| TBP-2 | TATA-binding protein |
| BCRP | breast cancer resistance protein |
| IUGR | intrauterine growth restriction |
| PE | preeclampsia |
| IL | interleukin |
| TNF | tumor necrosis factor |
| FABPs | fatty acid-binding proteins |
| IFNs | interferons |
| PGE2 | prostaglandin E2 |
| NF-κB | nuclear factor kappa-light-chain-enhancer of activated B cells |
| Syna | syncytin-A |
| Synb | syncytin-B |
| IGF | insulin-like growth factor |
| PAPP-A | pregnancy-associated plasma protein-A |
| IFGII | insulin-like growth factor |
| LOX | lysyl oxidase |
| MMP | matrix metalloproteinase |
| MEHP | mono ethylhexyl phthalate |
| TIMPs | tissue inhibitors of metalloproteinases |
| RUPP | uterine perfusion pressure |
| FATP | fatty acid transport protein |
| 9-HODE | 9S-hydroxy-10E,12Z-octadecadienoic acid |
| 13-HODE | 13S-hydroxy-9Z,11E-octadecadienoic acid |
| 15-HETE | 5S-hydroxy-5Z,8Z,11Z,13E-eicosatetraenoic acid |
| sFIt-1 | soluble fms-like tyrosine kinase-1 |
| uPA | urokinase plasminogen activator |
| PAI-1 | plasminogen activator inhibitor 1 |
| GDM | gestational diabetes mellitus |
References
- Brunmeir, R.; Xu, F. Functional Regulation of PPARs through Post-Translational Modifications. Int. J. Mol. Sci. 2018, 19, 1738. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bensinger, S.J.; Tontonoz, P. Integration of metabolism and inflammation by lipid-activated nuclear receptors. Nature 2008, 454, 470–477. [Google Scholar] [CrossRef] [PubMed]
- Tsukahara, T.; Matsuda, Y.; Haniu, H. Lysophospholipid-Related Diseases and PPARγ Signaling Pathway. Int. J. Mol. Sci. 2017, 18, 2730. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bogacka, I.; Kurzynska, A.; Bogacki, M.; Chojnowska, K. Peroxisome proliferator-activated receptors in the regulation of female reproductive functions. Folia Histochem. Cytobiol. 2015, 53, 189–200. [Google Scholar] [CrossRef]
- Holdsworth-Carson, S.J.; Lim, R.; Mitton, A.; Whitehead, C.; Rice, G.E.; Permezel, M.; Lappas, M. Peroxisome proliferator-activated receptors are altered in pathologies of the human placenta: Gestational diabetes mellitus, intrauterine growth restriction and preeclampsia. Placenta 2010, 31, 222–229. [Google Scholar] [CrossRef]
- Vitti, M.; Di Emidio, G.; Di Carlo, M.; Carta, G.; Antonosante, A.; Artini, P.G.; Cimini, A.; Tatone, C.; Benedetti, E. Peroxisome Proliferator-Activated Receptors in Female Reproduction and Fertility. PPAR Res. 2016, 2016, 4612306. [Google Scholar] [CrossRef]
- Pham, J.; Rajan, K.A.N.; Li, P.; Parast, M.M. The role of Sirtuin1-PPARγ axis in placental development and function. J. Mol. Endocrinol. 2018, 60, R201–R212. [Google Scholar] [CrossRef] [Green Version]
- Meher, A.; Sundrani, D.; Joshi, S. Maternal nutrition influences angiogenesis in the placenta through peroxisome proliferator activated receptors: A novel hypothesis. Mol. Reprod. Dev. 2015, 82, 726–734. [Google Scholar] [CrossRef]
- Barak, Y.; Sadovsky, Y.; Shalom-Barak, T. PPAR Signaling in Placental Development and Function. PPAR Res. 2008, 2008, 142082. [Google Scholar] [CrossRef] [Green Version]
- Wieser, F.; Waite, L.; Depoix, C.; Taylor, R.N. PPAR Action in Human Placental Development and Pregnancy and Its Complications. PPAR Res. 2008, 2008, 527048. [Google Scholar] [CrossRef] [Green Version]
- Desvergne, B.; Wahli, W. Peroxisome proliferator-activated receptors: Nuclear control of metabolism. Endocr. Rev. 1999, 20, 649–688. [Google Scholar]
- Tanaka, N.; Sugiyama, E.; Aoyama, T. Peroxisome proliferator-activated receptor (PPAR). Pharm. Res. 2001, 59 (Suppl. 2), 288–295. [Google Scholar]
- Dreyer, C.; Krey, G.; Keller, H.; Givel, F.; Helftenbein, G.; Wahli, W. Control of the peroxisomal beta-oxidation pathway by a novel family of nuclear hormone receptors. Cell 1992, 68, 879–887. [Google Scholar] [CrossRef]
- Kota, B.P.; Huang, T.H.; Roufogalis, B.D. An overview on biological mechanisms of PPARs. Pharm. Res. 2005, 51, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Kan, L.; Qi, C.; Kanwar, Y.S.; Yeldandi, A.V.; Rao, M.S.; Reddy, J.K. Isolation and characterization of peroxisome proliferator-activated receptor (PPAR) interacting protein (PRIP) as a coactivator for PPAR. J. Biol. Chem. 2000, 275, 13510–13516. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Escher, P.; Wahli, W. Peroxisome proliferator-activated receptors: Insight into multiple cellular functions. Mutagenesis Res. 2000, 448, 121–138. [Google Scholar] [CrossRef]
- Lalloyer, F.; Staels, B. Fibrates, glitazones, and peroxisome proliferator-activated receptors. Arter. Thromb. Vasc. Biol. 2010, 30, 894–899. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pawlak, M.; Lefebvre, P.; Staels, B. Molecular mechanism of PPARα action and its impact on lipid metabolism, inflammation and fibrosis in non-alcoholic fatty liver disease. J. Hepatol. 2015, 62, 720–733. [Google Scholar] [CrossRef] [Green Version]
- Derosa, G.; Sahebkar, A.; Maffioli, P. The role of various peroxisome proliferator-activated receptors and their ligands in clinical practice. J. Cell. Physiol. 2018, 233, 153–161. [Google Scholar] [CrossRef]
- Desvergne, B.; Michalik, L.; Wahli, W. Be fit or be sick: Peroxisome proliferator-activated receptors are down the road. Mol. Endocrinol. 2004, 18, 1321–1332. [Google Scholar] [CrossRef] [Green Version]
- Schmuth, M.; Haqq, C.M.; Cairns, W.J.; Holder, J.C.; Dorsam, S.; Chang, S.; Lau, P.; Fowler, A.J.; Chuang, G.; Moser, A.H.; et al. Peroxisome proliferator-activated receptor (PPAR)-beta/delta stimulates differentiation and lipid accumulation in keratinocytes. J. Investig. Dermatol. 2004, 122, 971–983. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Han, L.; Shen, W.J.; Bittner, S.; Kraemer, F.B.; Azhar, S. PPARs: Regulators of metabolism and as therapeutic targets in cardiovascular disease. Part II: PPAR-β/δ and PPAR-γ. Future Cardiol. 2017, 13, 279–296. [Google Scholar] [CrossRef] [PubMed]
- Ahmadian, M.; Suh, J.M.; Hah, N.; Liddle, C.; Atkins, A.R.; Downes, M.; Evans, R.M. PPARγ signaling and metabolism: The good, the bad and the future. Nat. Med. 2013, 19, 557–566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, S.; Dougherty, E.J.; Danner, R.L. PPARγ signaling and emerging opportunities for improved therapeutics. Pharm. Res. 2016, 111, 76–85. [Google Scholar] [CrossRef] [Green Version]
- Behl, T.; Kaur, I.; Goel, H.; Kotwani, A. Implications of the endogenous PPAR-gamma ligand, 15-deoxy-delta-12, 14-prostaglandin J2, in diabetic retinopathy. Life Sci. 2016, 153, 93–99. [Google Scholar] [CrossRef]
- Cardoso, S.; Santos, R.; Correia, S.; Carvalho, C.; Zhu, X.; Lee, H.G.; Casadesus, G.; Smith, M.A.; Perry, G.; Moreira, P.I. Insulin and Insulin-Sensitizing Drugs in Neurodegeneration: Mitochondria as Therapeutic Targets. Pharmaceuticals 2009, 2, 250–286. [Google Scholar] [CrossRef] [Green Version]
- Umemoto, T.; Fujiki, Y. Ligand-dependent nucleo-cytoplasmic shuttling of peroxisome proliferator-activated receptors, PPARα and PPARγ. Genes Cells 2012, 17, 576–596. [Google Scholar] [CrossRef]
- Burgermeister, E.; Seger, R. MAPK kinases as nucleo-cytoplasmic shuttles for PPARgamma. Cell Cycle 2007, 6, 1539–1548. [Google Scholar] [CrossRef] [Green Version]
- Benirschke, K.; Driscoll, S.G. The Pathology of the Human Placenta. In Placenta; Strauss, F., Benirschke, K., Driscoll, S.G., Eds.; Springer: Berlin/Heidelberg, Germany, 1967; pp. 97–571. [Google Scholar]
- Knöfler, M.; Haider, S.; Saleh, L.; Pollheimer, J.; Gamage, T.; James, J. Human placenta and trophoblast development: Key molecular mechanisms and model systems. Cell Mol. Life Sci. 2019, 76, 3479–3496. [Google Scholar] [CrossRef] [Green Version]
- Horii, M.; Touma, O.; Bui, T.; Parast, M.M. Modeling human trophoblast, the placental epithelium at the maternal fetal interface. Reproduction 2020, 160, R1–R11. [Google Scholar] [CrossRef]
- Turco, M.Y.; Moffett, A. Development of the human placenta. Development 2019, 146, 297. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Q.; Fujii, H.; Knipp, G.T. Expression of PPAR and RXR isoforms in the developing rat and human term placentas. Placenta 2002, 23, 661–671. [Google Scholar] [CrossRef] [PubMed]
- Schaiff, W.T.; Carlson, M.G.; Smith, S.D.; Levy, R.; Nelson, D.M.; Sadovsky, Y. Peroxisome proliferator-activated receptor-gamma modulates differentiation of human trophoblast in a ligand-specific manner. J. Clin. Endocrinol. Metab. 2000, 85, 3874–3881. [Google Scholar] [PubMed] [Green Version]
- Tarrade, A.; Schoonjans, K.; Pavan, L.; Auwerx, J.; Rochette-Egly, C.; Evain-Brion, D.; Fournier, T. PPARgamma/RXRalpha heterodimers control human trophoblast invasion. J. Clin. Endocrinol. Metab. 2001, 86, 5017–5024. [Google Scholar] [PubMed] [Green Version]
- Liu, F.; Rouault, C.; Guesnon, M.; Zhu, W.; Clément, K.; Degrelle, S.A.; Fournier, T. Comparative Study of PPARγ Targets in Human Extravillous and Villous Cytotrophoblasts. PPAR Res. 2020, 2020, 9210748. [Google Scholar] [CrossRef]
- Duan, S.Z.; Ivashchenko, C.Y.; Whitesall, S.E.; D’Alecy, L.G.; Duquaine, D.C.; Brosius, F.C., 3rd; Gonzalez, F.J.; Vinson, C.; Pierre, M.A.; Milstone, D.S.; et al. Hypotension, lipodystrophy, and insulin resistance in generalized PPARgamma-deficient mice rescued from embryonic lethality. J. Clin. Investig. 2007, 117, 812–822. [Google Scholar] [CrossRef] [Green Version]
- Tarrade, A.; Schoonjans, K.; Guibourdenche, J.; Bidart, J.M.; Vidaud, M.; Auwerx, J.; Rochette-Egly, C.; Evain-Brion, D. PPAR gamma/RXR alpha heterodimers are involved in human CG beta synthesis and human trophoblast differentiation. Endocrinology 2001, 142, 4504–4514. [Google Scholar] [CrossRef]
- Barak, Y.; Nelson, M.C.; Ong, E.S.; Jones, Y.Z.; Ruiz-Lozano, P.; Chien, K.R.; Koder, A.; Evans, R.M. PPAR gamma is required for placental, cardiac, and adipose tissue development. Mol. Cell 1999, 4, 585–595. [Google Scholar] [CrossRef]
- Nadeau, V.; Charron, J. Essential role of the ERK/MAPK pathway in blood-placental barrier formation. Development 2014, 141, 2825–2837. [Google Scholar] [CrossRef] [Green Version]
- Levytska, K.; Drewlo, S.; Baczyk, D.; Kingdom, J. PPAR- γ Regulates Trophoblast Differentiation in the BeWo Cell Model. PPAR Res. 2014, 2014, 637251. [Google Scholar] [CrossRef] [Green Version]
- Parast, M.M.; Yu, H.; Ciric, A.; Salata, M.W.; Davis, V.; Milstone, D.S. PPARgamma regulates trophoblast proliferation and promotes labyrinthine trilineage differentiation. PLoS ONE 2009, 4, e8055. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pavan, L.; Tarrade, A.; Hermouet, A.; Delouis, C.; Titeux, M.; Vidaud, M.; Thérond, P.; Evain-Brion, D.; Fournier, T. Human invasive trophoblasts transformed with simian virus 40 provide a new tool to study the role of PPARgamma in cell invasion process. Carcinogenesis 2003, 24, 1325–1336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Handschuh, K.; Guibourdenche, J.; Tsatsaris, V.; Guesnon, M.; Laurendeau, I.; Evain-Brion, D.; Fournier, T. Human chorionic gonadotropin produced by the invasive trophoblast but not the villous trophoblast promotes cell invasion and is down-regulated by peroxisome proliferator-activated receptor-gamma. Endocrinology 2007, 148, 5011–5019. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fournier, T.; Guibourdenche, J.; Handschuh, K.; Tsatsaris, V.; Rauwel, B.; Davrinche, C.; Evain-Brion, D. PPARγ and human trophoblast differentiation. J. Reprod. Immunol. 2011, 90, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Cocquebert, M.; Berndt, S.; Segond, N.; Guibourdenche, J.; Murthi, P.; Aldaz-Carroll, L.; Evain-Brion, D.; Fournier, T. Comparative expression of hCG β-genes in human trophoblast from early and late first-trimester placentas. Am. J. Physiol. Endocrinol. Metab. 2012, 303, E950–E958. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, M.; Lei, Z.M.; Rao Ch, V. The central role of human chorionic gonadotropin in the formation of human placental syncytium. Endocrinology 2003, 144, 1108–1120. [Google Scholar] [CrossRef] [Green Version]
- Muralimanoharan, S.; Kwak, Y.T.; Mendelson, C.R. Redox-Sensitive Transcription Factor NRF2 Enhances Trophoblast Differentiation via Induction of miR-1246 and Aromatase. Endocrinology 2018, 159, 2022–2033. [Google Scholar] [CrossRef]
- Handschuh, K.; Guibourdenche, J.; Cocquebert, M.; Tsatsaris, V.; Vidaud, M.; Evain-Brion, D.; Fournier, T. Expression and regulation by PPARgamma of hCG alpha- and beta-subunits: Comparison between villous and invasive extravillous trophoblastic cells. Placenta 2009, 30, 1016–1022. [Google Scholar] [CrossRef]
- Shalom-Barak, T.; Zhang, X.; Chu, T.; Timothy Schaiff, W.; Reddy, J.K.; Xu, J.; Sadovsky, Y.; Barak, Y. Placental PPARγ regulates spatiotemporally diverse genes and a unique metabolic network. Dev. Biol. 2012, 372, 143–155. [Google Scholar] [CrossRef] [Green Version]
- Daoud, G.; Simoneau, L.; Masse, A.; Rassart, E.; Lafond, J. Expression of cFABP and PPAR in trophoblast cells: Effect of PPAR ligands on linoleic acid uptake and differentiation. Biochim. Biophys. Acta 2005, 1687, 181–194. [Google Scholar] [CrossRef]
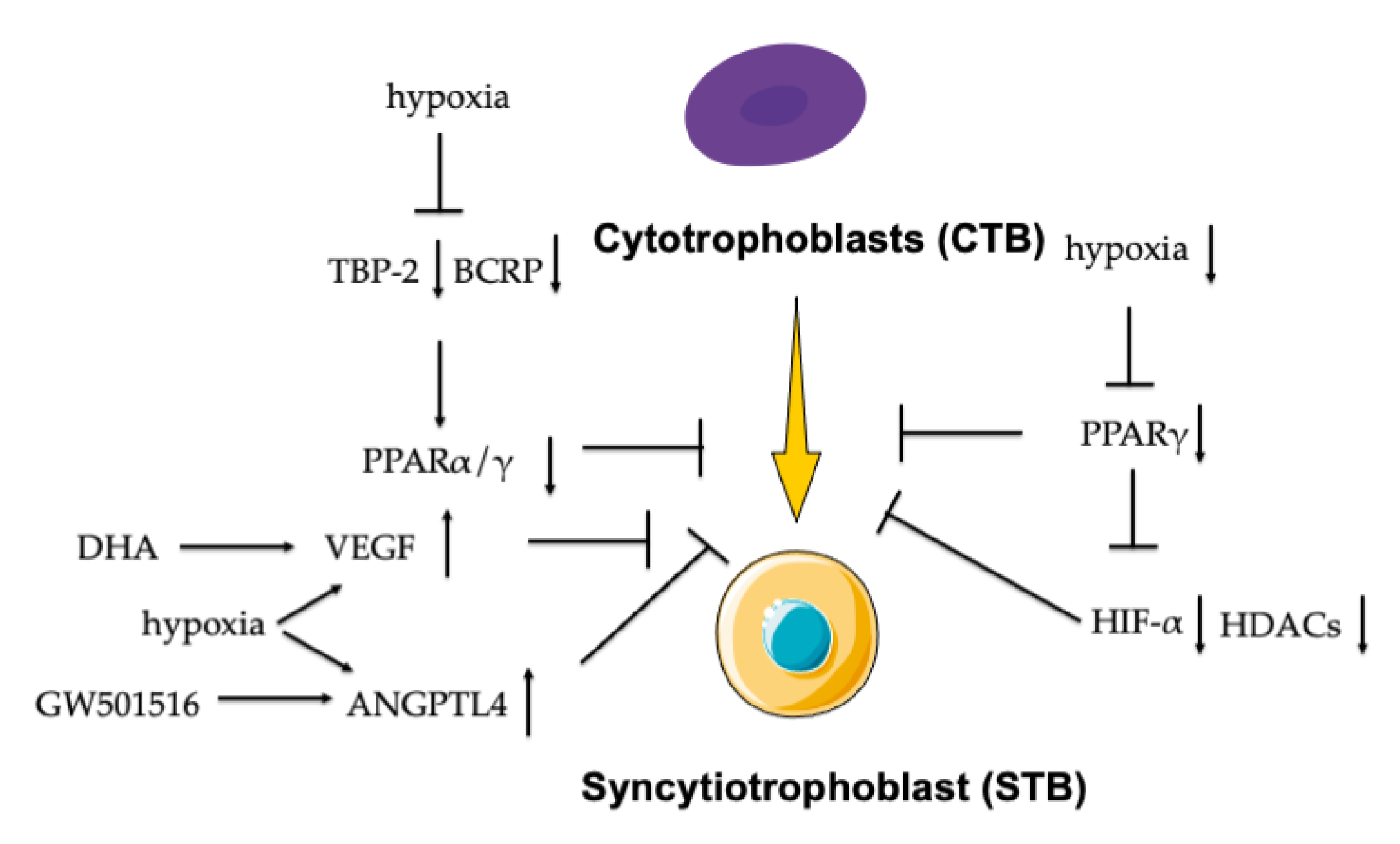
- Tache, V.; Ciric, A.; Moretto-Zita, M.; Li, Y.; Peng, J.; Maltepe, E.; Milstone, D.S.; Parast, M.M. Hypoxia and trophoblast differentiation: A key role for PPARγ. Stem Cells Dev. 2013, 22, 2815–2824. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wakeland, A.K.; Soncin, F.; Moretto-Zita, M.; Chang, C.W.; Horii, M.; Pizzo, D.; Nelson, K.K.; Laurent, L.C.; Parast, M.M. Hypoxia Directs Human Extravillous Trophoblast Differentiation in a Hypoxia-Inducible Factor-Dependent Manner. Am. J. Pathol. 2017, 187, 767–780. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cho, G.J.; Lee, L.H.; Lee, B.; Lee, J.; Ahn, K.H.; Hong, S.C.; Kim, H.J.; Oh, M.J. Effects of estradiol on HIF-1α expression and trophoblast differentiation in first trimester villous explant cultures. Obstet. Gynecol. Sci. 2018, 61, 71–78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Albers, R.E.; Kaufman, M.R.; Natale, B.V.; Keoni, C.; Kulkarni-Datar, K.; Min, S.; Williams, C.R.; Natale, D.R.C.; Brown, T.L. Trophoblast-Specific Expression of Hif-1α Results in Preeclampsia-Like Symptoms and Fetal Growth Restriction. Sci. Rep. 2019, 9, 2742. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olivo-Vidal, Z.E.; Rodríguez, R.C.; Arroyo-Helguera, O. Iodine Affects Differentiation and Migration Process in Trophoblastic Cells. Biol. Trace Elem. Res. 2016, 169, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Liao, T.L.; Chen, S.C.; Tzeng, C.R.; Kao, S.H. TCDD induces the hypoxia-inducible factor (HIF)-1α regulatory pathway in human trophoblastic JAR cells. Int. J. Mol. Sci. 2014, 15, 17733–17750. [Google Scholar] [CrossRef] [Green Version]
- Mogami, H.; Yura, S.; Kondoh, E.; Masutani, H.; Yodoi, J.; Konishi, I. Differential expression of thioredoxin binding protein-2/Txnip in human placenta: Possible involvement of hypoxia in its suppression during early pregnancy. J. Obstet. Gynaecol. Res. 2017, 43, 50–56. [Google Scholar] [CrossRef] [Green Version]
- Francois, L.N.; Gorczyca, L.; Du, J.; Bircsak, K.M.; Yen, E.; Wen, X.; Tu, M.J.; Yu, A.M.; Illsley, N.P.; Zamudio, S.; et al. Down-regulation of the placental BCRP/ABCG2 transporter in response to hypoxia signaling. Placenta 2017, 51, 57–63. [Google Scholar] [CrossRef] [Green Version]
- Johnsen, G.M.; Basak, S.; Weedon-Fekjær, M.S.; Staff, A.C.; Duttaroy, A.K. Docosahexaenoic acid stimulates tube formation in first trimester trophoblast cells, HTR8/SVneo. Placenta 2011, 32, 626–632. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, H.J.; Xia, X.R.; Diao, F.Y.; Ma, X.; Wang, J.; Gao, L.; Liu, J.; Gao, C.; Cui, Y.G.; et al. Hypoxia-induced and HIF1α-VEGF-mediated tight junction dysfunction in choriocarcinoma cells: Implications for preeclampsia. Clin. Chim. Acta 2019, 489, 203–211. [Google Scholar] [CrossRef]
- Burton, G.J.; Woods, A.W.; Jauniaux, E.; Kingdom, J.C. Rheological and physiological consequences of conversion of the maternal spiral arteries for uteroplacental blood flow during human pregnancy. Placenta 2009, 30, 473–482. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McMaster, M.T.; Zhou, Y.; Fisher, S.J. Abnormal placentation and the syndrome of preeclampsia. Semin. Nephrol. 2004, 24, 540–547. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, H.; Strauss, J.F., 3rd. Peroxisome proliferators and retinoids affect JEG-3 choriocarcinoma cell function. Endocrinology 1994, 135, 1135–1145. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Berga, S.L.; Zou, W.; Rajakumar, A.; Man, M.; Sidell, N.; Taylor, R.N. Human Endometrial Stromal Cell Differentiation is Stimulated by PPARβ/δ Activation: New Targets for Infertility? J. Clin. Endocrinol. Metab. 2020, 105, 2983–2995. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, R.; Kuroda, K.; Ikemoto, Y.; Ochiai, A.; Matsumoto, A.; Kumakiri, J.; Kitade, M.; Itakura, A.; Muter, J.; Brosens, J.J.; et al. Reprogramming of the retinoic acid pathway in decidualizing human endometrial stromal cells. PLoS ONE 2017, 12, e0173035. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Xie, H.; Sun, X.; Tranguch, S.; Zhang, H.; Jia, X.; Wang, D.; Das, S.K.; Desvergne, B.; Wahli, W.; et al. Stage-specific integration of maternal and embryonic peroxisome proliferator-activated receptor delta signaling is critical to pregnancy success. J. Biol. Chem. 2007, 282, 37770–37782. [Google Scholar] [CrossRef] [Green Version]
- Nadra, K.; Anghel, S.I.; Joye, E.; Tan, N.S.; Basu-Modak, S.; Trono, D.; Wahli, W.; Desvergne, B. Differentiation of trophoblast giant cells and their metabolic functions are dependent on peroxisome proliferator-activated receptor beta/delta. Mol. Cell. Biol. 2006, 26, 3266–3281. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Huo, Y.; He, W.; Liu, S.; Li, H.; Li, L. Visfatin is regulated by interleukin-6 and affected by the PPAR-γ pathway in BeWo cells. Mol. Med. Rep. 2019, 19, 400–406. [Google Scholar] [CrossRef]
- Blitek, A.; Szymanska, M. Peroxisome proliferator-activated receptor β/δ and γ agonists differentially affect prostaglandin E2 and cytokine synthesis and nutrient transporter expression in porcine trophoblast cells during implantation. Theriogenology 2020, 152, 36–46. [Google Scholar] [CrossRef]
- Zhang, Y.; Hu, L.; Cui, Y.; Qi, Z.; Huang, X.; Cai, L.; Zhang, T.; Yin, Y.; Lu, Z.; Xiang, J. Roles of PPARγ/NF-κB signaling pathway in the pathogenesis of intrahepatic cholestasis of pregnancy. PLoS ONE 2014, 9, e87343. [Google Scholar] [CrossRef]
- Pötgens, A.J.; Schmitz, U.; Bose, P.; Versmold, A.; Kaufmann, P.; Frank, H.G. Mechanisms of syncytial fusion: A review. Placenta 2002, 23 (Suppl. A), S107–S113. [Google Scholar] [CrossRef] [PubMed]
- Ruebner, M.; Langbein, M.; Strissel, P.L.; Henke, C.; Schmidt, D.; Goecke, T.W.; Faschingbauer, F.; Schild, R.L.; Beckmann, M.W.; Strick, R. Regulation of the human endogenous retroviral Syncytin-1 and cell-cell fusion by the nuclear hormone receptors PPARγ/RXRα in placentogenesis. J. Cell Biochem. 2012, 113, 2383–2396. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.; Zhang, Y.; Liu, L.; Yuan, H.; Qu, J.; Shen, R. Activation of peroxisome proliferator activator receptor delta in mouse impacts lipid composition and placental development at early stage of gestation. Biol. Reprod. 2014, 91, 57. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhuang, X.; Jiang, M.; Guan, F.; Fu, Q.; Lin, J. ANGPTL4 mediates the protective role of PPARγ activators in the pathogenesis of preeclampsia. Cell Death Dis. 2017, 8, e3054. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mayama, R.; Izawa, T.; Sakai, K.; Suciu, N.; Iwashita, M. Improvement of insulin sensitivity promotes extravillous trophoblast cell migration stimulated by insulin-like growth factor-I. Endocr. J. 2013, 60, 359–368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Workalemahu, T.; Enquobahrie, D.A.; Gelaye, B.; Thornton, T.A.; Tekola-Ayele, F.; Sanchez, S.E.; Garcia, P.J.; Palomino, H.G.; Hajat, A.; Romero, R.; et al. Abruptio placentae risk and genetic variations in mitochondrial biogenesis and oxidative phosphorylation: Replication of a candidate gene association study. Am. J. Obstet. Gynecol. 2018, 219, e1–e617. [Google Scholar] [CrossRef]
- Fournier, T.; Pavan, L.; Tarrade, A.; Schoonjans, K.; Auwerx, J.; Rochette-Egly, C.; Evain-Brion, D. The role of PPAR-gamma/RXR-alpha heterodimers in the regulation of human trophoblast invasion. Ann. N. Y. Acad. Sci. 2002, 973, 26–30. [Google Scholar] [CrossRef]
- Handschuh, K.; Guibourdenche, J.; Guesnon, M.; Laurendeau, I.; Evain-Brion, D.; Fournier, T. Modulation of PAPP-A expression by PPARgamma in human first trimester trophoblast. Placenta 2006, 27 (Suppl. A), S127–S134. [Google Scholar] [CrossRef] [Green Version]
- Segond, N.; Degrelle, S.A.; Berndt, S.; Clouqueur, E.; Rouault, C.; Saubamea, B.; Dessen, P.; Fong, K.S.; Csiszar, K.; Badet, J.; et al. Transcriptome analysis of PPARγ target genes reveals the involvement of lysyl oxidase in human placental cytotrophoblast invasion. PLoS ONE 2013, 8, e79413. [Google Scholar] [CrossRef] [Green Version]
- Li, S.J.; Shang, T.; Li, S.Y.; Li, Q.L. Effects of peroxisome proliferator-activated receptor gamma and its ligands on cytotrophoblast invasion in first trimester of pregnancy and mechanism thereof. Zhonghua Yi Xue Za Zhi 2007, 87, 174–178. [Google Scholar]
- Kolben, T.M.; Rogatsch, E.; Vattai, A.; Hester, A.; Kuhn, C.; Schmoeckel, E.; Mahner, S.; Jeschke, U.; Kolben, T. PPARγ Expression Is Diminished in Macrophages of Recurrent Miscarriage Placentas. Int. J. Mol. Sci. 2018, 19, 1872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knabl, J.; Vattai, A.; Hüttenbrenner, R.; Hutter, S.; Karsten, M.; Jeschke, U. RXRα is upregulated in first trimester endometrial glands of spontaneous abortions unlike LXR and PPARγ. Eur. J. Histochem. 2016, 60, 2665. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, F.; Hu, W.; Li, Y.; Shen, H.; Hu, J. Mono-2-ethylhexyl phthalate inhibits human extravillous trophoblast invasion via the PPARγ pathway. Toxicol. Appl. Pharm. 2017, 327, 23–29. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, F.P.; Drewlo, S.; Kingdom, J.; Johns, E.J.; Walsh, S.K.; Kenny, L.C. Peroxisome proliferator-activated receptor-γ as a potential therapeutic target in the treatment of preeclampsia. Hypertension 2011, 58, 280–286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dubé, E.; Gravel, A.; Martin, C.; Desparois, G.; Moussa, I.; Ethier-Chiasson, M.; Forest, J.C.; Giguère, Y.; Masse, A.; Lafond, J. Modulation of fatty acid transport and metabolism by maternal obesity in the human full-term placenta. Biol. Reprod. 2012, 87, 14. [Google Scholar] [CrossRef]
- Bildirici, I.; Roh, C.R.; Schaiff, W.T.; Lewkowski, B.M.; Nelson, D.M.; Sadovsky, Y. The lipid droplet-associated protein adipophilin is expressed in human trophoblasts and is regulated by peroxisomal proliferator-activated receptor-gamma/retinoid X receptor. J. Clin. Endocrinol. Metab. 2003, 88, 6056–6062. [Google Scholar] [CrossRef] [Green Version]
- Mariani, T.J.; Budhraja, V.; Mecham, B.H.; Gu, C.C.; Watson, M.A.; Sadovsky, Y. A variable fold change threshold determines significance for expression microarrays. FASEB J. 2003, 17, 321–323. [Google Scholar] [CrossRef] [Green Version]
- Schaiff, W.T.; Bildirici, I.; Cheong, M.; Chern, P.L.; Nelson, D.M.; Sadovsky, Y. Peroxisome proliferator-activated receptor-gamma and retinoid X receptor signaling regulate fatty acid uptake by primary human placental trophoblasts. J. Clin. Endocrinol. Metab. 2005, 90, 4267–4275. [Google Scholar] [CrossRef] [Green Version]
- Díaz, P.; Harris, J.; Rosario, F.J.; Powell, T.L.; Jansson, T. Increased placental fatty acid transporter 6 and binding protein 3 expression and fetal liver lipid accumulation in a mouse model of obesity in pregnancy. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015, 309, R1569–R1577. [Google Scholar] [CrossRef] [Green Version]
- Tian, L.; Wen, A.; Dong, S.; Yan, P. Molecular Characterization of Microtubule Affinity-Regulating Kinase4 from Sus scrofa and Promotion of Lipogenesis in Primary Porcine Placental Trophoblasts. Int. J. Mol. Sci. 2019, 20, 1206. [Google Scholar] [CrossRef] [Green Version]
- Schild, R.L.; Schaiff, W.T.; Carlson, M.G.; Cronbach, E.J.; Nelson, D.M.; Sadovsky, Y. The activity of PPAR gamma in primary human trophoblasts is enhanced by oxidized lipids. J. Clin. Endocrinol. Metab. 2002, 87, 1105–1110. [Google Scholar] [PubMed] [Green Version]
- Cawyer, C.R.; Horvat, D.; Leonard, D.; Allen, S.R.; Jones, R.O.; Zawieja, D.C.; Kuehl, T.J.; Uddin, M.N. Hyperglycemia impairs cytotrophoblast function via stress signaling. Am. J. Obstet. Gynecol. 2014, 211, e1–e8. [Google Scholar] [CrossRef] [PubMed]
- Cawyer, C.; Afroze, S.H.; Drever, N.; Allen, S.; Jones, R.; Zawieja, D.C.; Kuehl, T.; Uddin, M.N. Attenuation of hyperglycemia-induced apoptotic signaling and anti-angiogenic milieu in cultured cytotrophoblast cells. Hypertens Pregnancy 2016, 35, 159–169. [Google Scholar] [CrossRef]
- Jones, H.N.; Jansson, T.; Powell, T.L. Full-length adiponectin attenuates insulin signaling and inhibits insulin-stimulated amino Acid transport in human primary trophoblast cells. Diabetes 2010, 59, 1161–1170. [Google Scholar] [CrossRef] [Green Version]
- Aye, I.L.; Powell, T.L.; Jansson, T. Review: Adiponectin--the missing link between maternal adiposity, placental transport and fetal growth? Placenta 2013, 34 (Suppl. S40), 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
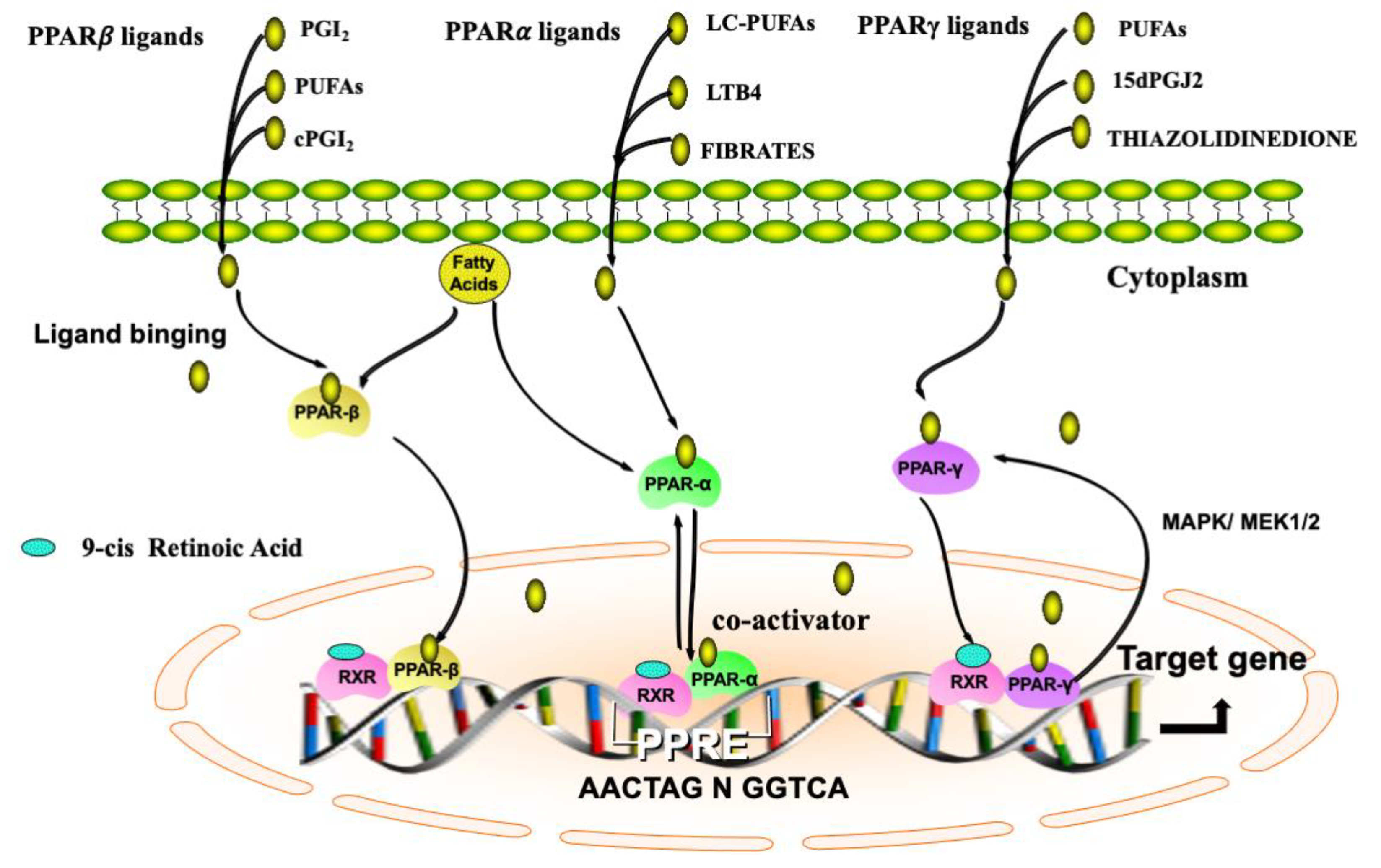

| Receptor | Natural Ligands | Synthetic Ligands |
|---|---|---|
| PPARα | arachidonic acid | fibrates |
| linoleic acid | industrial plasticizers | |
| leukotriene B4 | naveglitazar | |
| docosahexaenoic acid | netoglitazone | |
| eicosapentaenoic acid | muraglitazar | |
| prostaglandin I2 (PGI2) | ||
| PPARβ/δ | polyunsaturated fatty acids (PUFAs) | carbaprostacyclin (cPGI2) |
| eicosanoids | iloprost | |
| PPARγ | fatty acids | rosiglitazone |
| oxidized low-density lipoprotein | thiazolidinedione | |
| 15-deoxy-12,14 prostaglandin J2 (15dPGJ2), | ciglitazone | |
| prostaglandin D2 | troglitazone | |
| 13- hydroxyoctadecadienoic acid (HODE) | GW1929 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, L.; Yang, H.; Ye, Y.; Ma, Z.; Kuhn, C.; Rahmeh, M.; Mahner, S.; Makrigiannakis, A.; Jeschke, U.; von Schönfeldt, V. Role of Peroxisome Proliferator-Activated Receptors (PPARs) in Trophoblast Functions. Int. J. Mol. Sci. 2021, 22, 433. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22010433
Peng L, Yang H, Ye Y, Ma Z, Kuhn C, Rahmeh M, Mahner S, Makrigiannakis A, Jeschke U, von Schönfeldt V. Role of Peroxisome Proliferator-Activated Receptors (PPARs) in Trophoblast Functions. International Journal of Molecular Sciences. 2021; 22(1):433. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22010433
Chicago/Turabian StylePeng, Lin, Huixia Yang, Yao Ye, Zhi Ma, Christina Kuhn, Martina Rahmeh, Sven Mahner, Antonis Makrigiannakis, Udo Jeschke, and Viktoria von Schönfeldt. 2021. "Role of Peroxisome Proliferator-Activated Receptors (PPARs) in Trophoblast Functions" International Journal of Molecular Sciences 22, no. 1: 433. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22010433





